One of the major health problems is pain. Studies indicate that 20% of the general population in both the USA and Europe are suffering from long-term pain. As per the International Association for the Study of Pain (IASP), pain is defined as ‘an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage’.
So far, managing chronic pain by way of the clinic has seen very limited progress in recent decades. Pain relief treatment has so far largely relied on predominantly two classical medications: opioids and non-steroidal anti-inflammatory drugs (NSAIDs), and is still dominated by these. However, these classical medications suffer from a few drawbacks, including side effects such as dependence (in the case of opioids) and gastric ulceration (prevalent in the case of NSAIDs). This demonstrates the urgent need for new drug targets as well as novel compounds to usher in a new era of pain therapeutics.
The three types of classification of pain are nociceptive pain, inflammatory pain, and neuropathic or pathological pain. The first category i.e. nociceptive pain involves nociceptors, which are peripheral sensory nerve fibers that transduce painful stimuli. In contrast, inflammatory pain involves the immunological response to injury through inflammatory mediators contributing to pain. Finally, neuropathic or pathological pain usually is a result of damage or disease affecting the sensory system.
As per the latest pain research guide, there is an enhanced focus on nociception and the transduction of pain to the spinal cord. This guide examines some of the primary classical targets – in addition to pain targets that are emerging. It can be expected that a thorough understanding of nociceptive pain will enable the identification of key interventions that are most likely to provide therapeutic benefit in the future. Moreover, Tocris Bioscience provides a range of high-performance life science reagents, enabling researchers to target the mechanisms of underlying pain.
Nociception
The process that involves transduction of intense thermal, chemical, or mechanical stimuli detected by a subpopulation of peripheral nerve fibers (nociceptors aka pain receptors) is known as nociception. As described above, while neuropathic pain is a result of damage or disease that affects the sensory system, nociceptive pain, in contrast, is the body’s normal response to noxious insult or injury of tissues, including the skin, muscle, organs, joints, tendons, and bones.
Furthermore, inflammatory pain – though not discussed in this guide – is characterized by white blood cells and antibodies being mobilized, thus leading to swelling and fluid accumulation. Inflammatory mediators can enhance or sensitize noxious signals, leading to the activation of pain fibers by this lower intensity stimuli. In such a scenario, the pain generated can be more persistent.
Medications such as NSAIDs (aspirin, ibuprofen, and valdecoxib) inhibit cyclooxygenases, making them among the most popular drug types used in the prevention of inflammatory pain. However, these drug types are limited by some severe side effects, highlighting the need for new pain targets.
Activated by noxious stimuli such as tissue injury or exposure to an acid or irritant, nociceptors can also be brought about by extreme temperatures. Here, electrophysiological activity is generated, which is then transmitted to the spinal cord. Functionally, nociceptors are capable of being divided into three primary compartments: the peripheral terminal (responsible for detecting painful stimuli); the axon (aiding transduction of the signal); and the presynaptic terminal (involved in transmitting the signal, using glutamate as a primary neurotransmitter, across the synapse to second-order neurons).
There are two main categories of nociceptors: myelinated A-fibers and unmyelinated C-fibers. The speed of transmission correlates to the axon diameter of sensory neurons depending on whether they are myelinated. Most nociceptors are unmyelinated C-fibers contributing to a poorly localized sensation of secondary pain. In contrast, fast-onset, a sharp pain is mediated by myelinated A-fibers.
In the dorsal root ganglion (DRG), the cell bodies of nociceptive neurons send two processes: one axon to the peripheral tissue, and a second axon that synapses on second-order neurons in the dorsal horn of the spinal cord. Entering the spinal cord via the dorsal root, the central axon of DRG neurons branches to innervate multiple spinal segments in the rostral and caudal direction (laminae I, II, IIA, and V), from which the ascending nociceptive pathways originate (Figure 1). DRG neurons may interact with both excitatory and inhibitory interneurons within these laminae, helping to fine-tune the incoming signals.
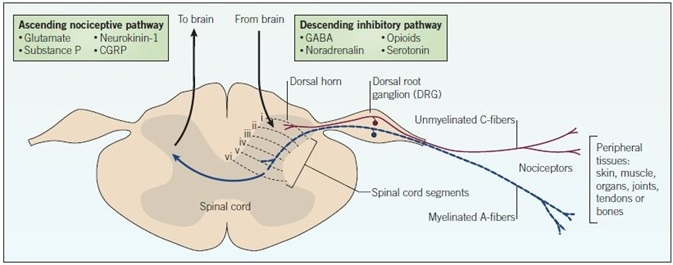
Figure 1. Nociceptive pain pathway. The cell bodies of nociceptors are located in the dorsal root ganglion (DRG) and terminate as free endings in peripheral tissues. Pain signals originating from the periphery pass through the dorsal root ganglion carried by C-fiber nerves (red) and myelinated A-fiber nerves (blue). Inputs directed to the dorsal horn synapse on interneurons that modulate the transmission of nociceptive signals to higher CNS centers. Signals are relayed to the brain via ascending pathways, and descending pathways from the brain send inhibitory signals. Highlighted in the boxes are key mediators and drug targets that play important roles in pain processing and transmission.
A key role is played by calcium channels in the transmission of the pain signal, due to the triggered release of neuropeptides such as substance P, neurokinin 1, and calcitonin gene-related peptide (CGRP), in addition to neurotransmitters such as glutamate. After that, the ascending relay neurons then project to the medulla, mesencephalon, and thalamus regions in the brain, which in turn project to the somatosensory and anterior cingulate cortices to drive the cognitive aspects of pain.
Both local GABA-releasing inhibitory interneurons in the dorsal horn as well as descending noradrenergic neurons that originate in the brain can inhibit pain signaling. Signals relayed from nociceptors can potentially act in combination, producing changes that lead to hyperalgesia (which refers to an exaggerated response to normally painful mechanical or thermal stimuli), or allodynia (i.e. pain from a stimulus that would not normally provoke pain).
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.