Atherosclerosis is a disease occurring in the arterial system that is distinguished by the build-up of fatty deposits within arterial walls (Figure 2). These fatty deposits referred to as ‘atherosclerotic plaques’, also comprise cellular debris and, among advanced plaques, often calcium deposits (in the form of hydroxyapatite). The plaque contents are extremely thrombogenic and are thus separated from the circulating blood by a fibrous cap. This is primarily constituted by vascular smooth muscle cells and extracellular matrix proteins, such as collagen, which undergo synthesis by these cells (Figure 1).
What Is Atherosclerosis?
Although the precise cause of atherosclerosis continues to elude researchers, the initial mechanism – known as ‘atherogenesis’ – is understood. Atherogenesis comprises the transmigration of macrophages/monocytes across the endothelium into the intima, wherein they scavenge oxidized lipid (ox-LDL), forming lipid-laden ‘foam cells’. This transmigration is promoted by the expression of adhesion molecules by endothelial cells. Foam cells undergo apoptosis, establishing a lipid-rich core in the developing plaque. The establishment of a plaque in the arterial wall activates an inflammatory response, spread by the secretion of growth factors and chemokines by resident plaque cells.
Circulating macrophages, recruited to the atherosclerotic plaque via chemotaxis, are an important source of matrix metalloproteinases (MMPs). These zinc-dependent enzymes cause degradation of the fibrous cap, which predisposes the plaque to rupture. Moreover, they can also cause degradation of the elastic laminae between layers of the arterial wall, which facilitates the movement of smooth muscle cells from the tunica media to the fibrous cap, equipping it with extra strength.
A thick smooth muscle-cell rich fibrous cap embodying a small lipid core is typical of a ‘stable’ plaque, which refers to a plaque less susceptible to rupture. Plaques embodying a large, necrotic lipid core and a thin fibrous cap are known as ‘vulnerable’ or ‘unstable’ plaques, which are more likely to rupture. All defects in the fibrous cap expose the thrombogenic plaque contents to the circulating blood, triggering thrombosis. Although 90% of these plaque ruptures are clinically silent, the remaining percentage lead to myocardial infarction or stroke and can be fatal. As well as the recruitment of macrophages in an atherosclerotic plaque, additional immune mediators also occur during atherosclerosis.
The expression of several pro-atherogenic cytokines, comprising interleukin (IL)-1 and IL-6, are understood to be upregulated among atherosclerotic plaques. Consequently, targeting the receptors of these cytokines might inhibit the progression of atherosclerosis. While triggering the immune response in an atherosclerotic plaque might prove disadvantageous, as it drives the progression and eventual rupture of a plaque, it may equally prove beneficial in resolving the inflammation and clearing necrotic foam cells from the core of the plaque. In this way, targeting the immune response in atherosclerosis can represent a ‘double-edged sword’.
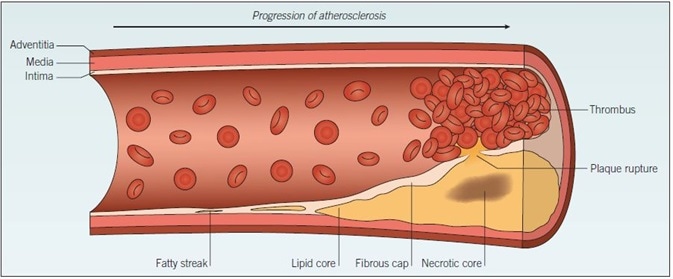
Figure 1. Atherosclerosis. The development of atherosclerosis within a blood vessel wall starts as a fatty streak, composed of a small cluster of lipid-laden macrophages, which progresses over time to form a lipid pool within the vessel wall. Cell debris from apoptotic foam cells within the lipid pool forms a necrotic core. The fibrous cap separates the lipid pool from the blood, but the rupture of this cap exposes the thrombogenic plaque contents to circulating blood, triggering thrombus formation.
Figure 2. Section through a coronary artery with an atherosclerotic plaque. Lipid accumulation and inflammatory cell invasion (depicted in yellow) within an arterial wall (pink) cause vessel narrowing and stenosis. A thin fibrous cap (arrows) separates the plaque contents from the circulating blood. Rupture of the fibrous cap exposes these thrombogenic contents to the blood, triggering thrombosis.
Therapeutics in Atherosclerosis
The primary objective of contemporary therapeutics in atherosclerosis is to lower blood cholesterol, thus restricting the expansion of an atherosclerotic plaque. The exemplary lipid-lowering drugs are the statins, although additional compounds comprising CI 976, probucol, and torcetrapib also display antihypercholesterolemic behavior. However, their side effect profiles restrict their usage in the clinic. In addition to lipid-lowering, the coagulation cascade represents an additional potential direction in atherosclerosis therapy. This is because it is the formation of an occlusive thrombus that activates the pathologies associated with atherosclerosis, rather than plaque rupture per se.
Other Mediators
Possible targets within the coagulation cascade comprise tissue plasminogen activator (tPA), urokinase plasminogen activator (uPA), and thrombin. Other mediators included in platelet activation represent further possible therapeutic targets in the prevention and treatment of atherothrombosis. Further targets embodying therapeutic potential in atherosclerosis comprise urotensin II and phospholipases, as the occurrence of both can trigger foam cell formation, which furthers the development of atherosclerosis.
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.