Although thrombosis is an essential hemostatic process for the prevention of excessive blood loss in the aftermath of an injury, aberrant thrombosis can activate pathological conditions such as myocardial infarction and stroke. As a result, the initiation of thrombosis is strictly regulated under physiological conditions. Platelets, the production of which occurs in the bone marrow, are anucleate cell fragments of megakaryocytes. They represent a key element of thrombosis, exhibiting a rapid, exponential activation in the event of tissue damage.
Different Categories of Granules
Although they do not have a nucleus, platelets possess two different categories of granules within the cytoplasm – alpha granules and dense granules – while also expressing several different receptors on their plasma membranes (Figure 1). Alpha and dense granules each comprise an assortment of bioactive mediators, such as 5-HT, ADP, and calcium, in addition to growth factors including insulin-like growth factor-1 (IGF-1), platelet-derived growth factor (PDGF) and transforming growth factor (TGF) β1. Following activation, platelets undergo degranulation, releasing granule contents into the surrounding environment, and promoting the activation and aggregation of contiguous platelets. Receptors present on the platelet plasma membrane comprise the 5-HT receptor 5-HT2A; the thrombin receptors (protease-activated receptors) PAR1 and PAR4; the thromboxane A2 (TXA2) receptor TP; and the purinergic receptors P2X1, P2Y1, and P2Y12.
Platelet Signaling
Platelet signaling can also undergo activation following exposure to collagen via the glycoprotein receptor, GPVI. Important downstream mediators of these receptors comprise diacylglycerol (DAG) and the plasma membrane phospholipid phosphatidylinositol 4,5-bisphosphate (PIP2), which is split by phospholipase C to form inositol triphosphate (IP3). IP3 receptor (InsP3R) activation stimulates the efflux of calcium ions from intracellular calcium stores, such as the endoplasmic reticulum, precipitating an increase in intracellular calcium (Figure 1).
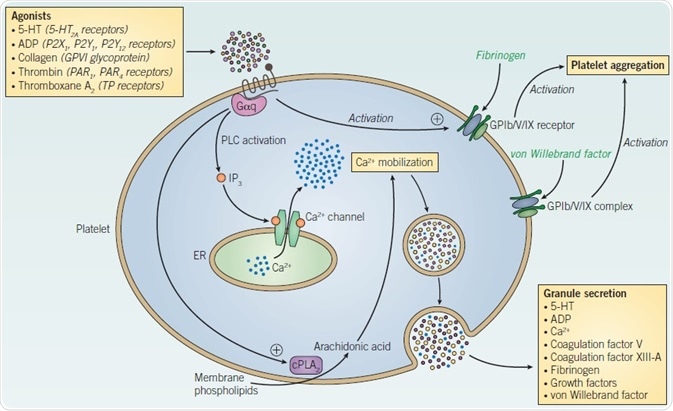
Figure 1. Platelet signaling and activation.
Agonist stimulation of platelets triggers activation of glycoprotein Ib/V/IX; production of arachidonic acid from membrane phospholipids by the actions of cytosolic phospholipase A2 (cPLA2); and mobilization of intracellular calcium. This consequently engenders the release of granules containing platelet-activating mediators, which in turn act on contiguous platelets, augmenting platelet activation and aggregation.
The mobilization of calcium triggers the exocytosis of cytoplasmic granules and the activation of platelet aggregation-inducing glycoproteins, including GPIIb/IIIa. GPIIb/IIIa acts as a cell surface receptor for fibrinogen, promoting platelet activation.
The central role of GPIIb/IIIa in platelet aggregation makes it a useful therapeutic target in anticoagulant therapy. GPIIb/IIIa inhibitors, including echistatin and abciximab, are contemporarily utilized as prophylactic therapy during angioplasty to obstruct thrombus formation. Inhibitors of P2Y12 – such as clopidogrel and ticlodipine– likewise indirectly obstruct platelet aggregation by inhibiting the activation of GPIIb/IIIa. In addition to GPIIb/IIIa, the integrin receptor complex GPIb/ V/IX is also essential in thrombosis. Platelet adhesion is promoted via interaction of the GPIb/V/IX complex with von Willebrand factor (vWF), which occurs on exposed collagen at the site of vascular damage. This is accomplished via GPIb/V/IX-mediated phosphorylation of intracellular signaling proteins, comprising Fc receptor γ-chain (FcRγ), PLCγ and pp60Src, in turn initiating and augmenting the adhesion of platelets with neighboring platelets and also the extracellular matrix.
Phosphorylation of FcRγ by GPIb/V/IX instigates FcRγ-GPIb/V/IX functional coupling, thus promoting platelet activation via immunoreceptor tyrosine-based activation motif (ITAM)-driven signaling. Agonist stimulation of platelets also instigates the generation of arachidonic acid from membrane phospholipids, a reaction which undergoes catalysis by cytosolic phospholipase A2 (cPLA2). Arachidonic acid can be used by both lipoxygenases (LOX), in order to form the lipid mediator hydroperoxyeicosatetraenoic acid (HPETE), and by cyclooxygenases (COX), in order to form prostaglandin H2 (PGH2).
Inhibitors as Effective Therapeutic Strategy
PGH2 undergoes further metabolization to form prothrombotic eicosanoids, such as TXA2. Inhibition of the PGH2 synthesis pathway utilizing COX inhibitors is an effective therapeutic strategy for preventing thrombosis. These inhibitors comprise the TXA2 synthase inhibitor, dipyridamole, aspirin, celecoxib, and diclofenac. PLA2 inhibitors, such as AACOCF3, also display antithrombotic effects via inhibition of the activity of the arachidonic acid pathway.
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.