G-Protein Coupled Receptors (GPCRs) are seven transmembrane domain receptors with the ability to move between active-like (R* and R**) and inactive-like (R, R′, and R″) states. This may take place in the absence of transducer or ligand (the Apo state), but the energy barrier to achieving and maintaining active-like conformations implies that it is not likely to happen or has a lower probability in the absence of an agonist and G protein (or another transducer).
The active GPCR conformation is stabilized by the presence of both a transducer (for example, G proteins) and agonist, thereby offering the maximum probability for a signaling event to occur. The rates of turnover and transducer engagement are modified in an agonist-dependent way that ascertains the observed efficacy1. GPCRs can also communicate with regulatory proteins or with several distinct transducers, and these can be preferably involved in an agonist-specific manner, which results in biased agonism1. Nanobodies that attach to the GPCRs’ intracellular face can allosterically drive either active- or inactive-like conformations and have been used in structural studies to capture these states2; however, larger numbers of G proteins-containing structures are being solved at present3.
GPCRs are pleiotropically connected and therefore can associate with many different G proteins (Figure 1) or other protein transducers (Figure 2) for regulating intracellular signaling.
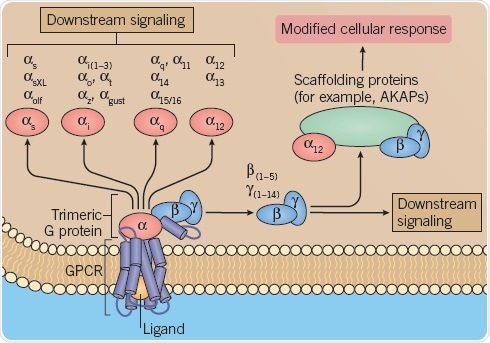
Fig 1. G-protein activation. Image credit: Tocris Bioscience
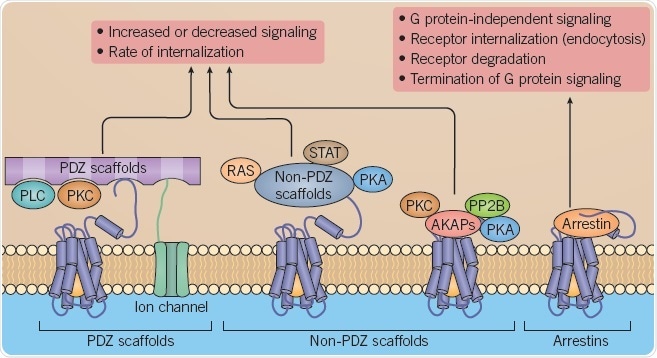
Fig 2. B: Scaffolding proteins regulating GPCR signaling. Image credit: Tocris Bioscience
Multiple Mechanisms Elicit Biased Agonism
Within GPCRs, different conformations are induced by different ligands, thereby promoting binding conditions for specific transducer proteins like arrestins and G proteins (Figure 3).
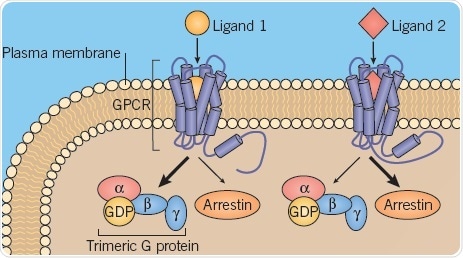
Fig 3. Different GPCR conformations. Image credit: Tocris Bioscience
Within scaffolding proteins such as arrestins, particular ligand-induced receptor conformations induce different conformational variations, which activate different downstream signaling pathways (Figure 4).
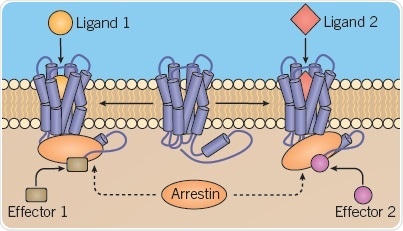
Fig 4. B: Different scaffolding protein conformations. Image credit: Tocris Bioscience
The rate of GTP–GDP exchange can be controlled by various ligands that induce different conformational rearrangements within G proteins. Ligands that promote rapid G protein turnover and GTP association enable the occurrence of more signaling events than the ligands that induce a slow rate of exchange (Figure 5).
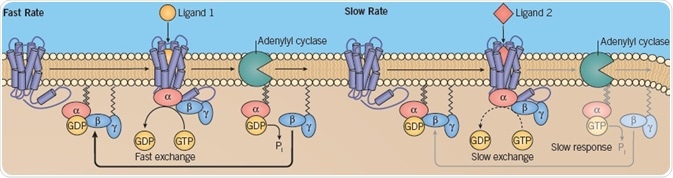
Fig 5. Different rate of GTP–GDP exchange. Image credit: Tocris Bioscience
Identifying Biased Agonism at the Adenosine A1 GPCR (A1R) Using the Operational Model
The activation of different signaling events for three A1R agonists — VCP746 (purple line), R-PIA (orange line), and NECA (green line)4 — is demonstrated in the six concentration-response curves (Figure 6). The transduction ratio (logτ/KA; connecting agonist occupied receptors to pharmacological effects) was derived by using operational modeling, and this was plotted in a “Web of Bias”5.
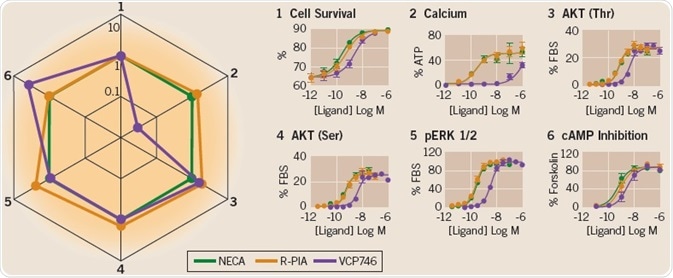
Fig 6. The Web of Bias. Image credit: Tocris Bioscience
Each drug profile with respect to the reference pathway 1 and reference agonist NECA is illustrated in the “Web of Bias” chart (as ∆∆logτ/KA). The biased agonism of VCP746 for pathway 2 (reduced response with respect to pathway 1 and NECA) can be easily seen using this visual representation. R-PIA shows a similar signaling profile compared to the reference agonist, NECA.
Importantly, biased agonism is relative at all times and must be expressed in comparison to a reference ligand. The observations of bias can vary in a temporal and system (cell background)-dependent manner1.
Spatial and Kinetic Factors
Biased agonism is controlled by the temporal stability of biomolecular interactions between receptor, ligand, and regulatory proteins, or transducers. Using biased agonism, different ligands can modify the location, strength, and type of signaling through one receptor type (Figure 7).
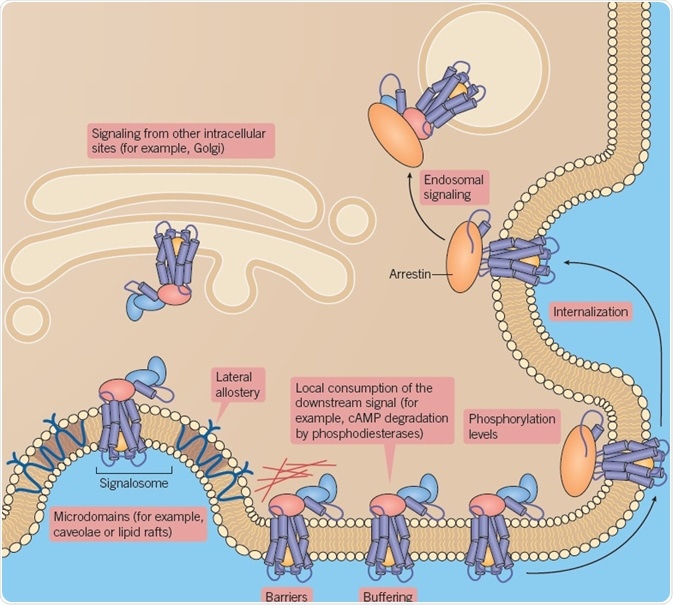
Fig 7. Biased agonism controlled by biomolecular interactions. Image credit: Tocris Bioscience
Therapeutic Application of Biased Agonism
The efficacy and safety profiles of drugs can be improved by examining the pharmacology of GPCR biased agonists. Patient outcomes and translation of drugs to the clinic can be optimized by identifying compounds with a therapeutically advantageous activation profile with reduced undesired on-target signaling. Yet, an enhanced knowledge of the patterns of tissue-specific and integrated signaling is required for the selection of the most suitable biased agonists as it mediates several outcomes physiologically and also in a disease-specific context.
Altogether, such improvement in understanding of disease along with improvement in GPCR structure determination, drug design, and pharmacological assessment of compounds should offer a large range of new biased agonist drugs to the clinic.
References
- Wootten et al. (2018) Nat Rev Mol Cell Biol 19:638–653
- Thal et al. (2018) Nature 559:45–53
- Glukhova et al. (2018) ACS Pharmacol Transl Sci 1:73–83
- Baltos et al. (2016) Biochem Pharmacol 99:101–112
- Kenakin et al. (2012) ACS Chem Neurosci 3:193–203
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.