The term "addiction" is often controversial because it can be both divisive and stigmatizing; however, in this article, addiction is used as a valuable shorthand for the processes through which people become reliant on the use of drugs, or activities like gambling, to the point where it distorts and even takes over their lives.
The significant feature of drug addiction is the inability to discontinue the use of drugs in spite of clear evidence of their harmful effects. The drug becomes increasingly more important, controlling life at the cost of work, friends, and family. Not everyone who uses drugs becomes addicted and factors that cause addiction include craving, class of drug, and family history of addiction and propensity to withdrawal reactions. Specifically, using drugs to self-medicate psychiatric issues is likely to cause addiction.
Brain Circuits Associated with Addiction
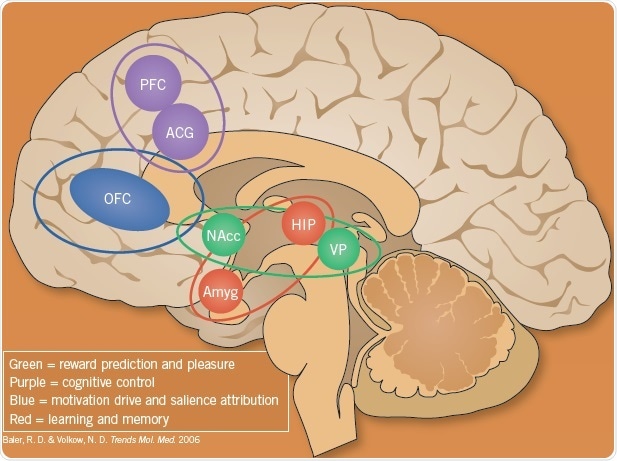
Fig 1. The brain circuits involved in addition. Image credit: Tocris Bioscience
The brain circuits that are involved in addiction have been characterized in preclinical studies and in human imaging studies. The four interacting circuits that can be involved, to varying extents, in different persons and different addictions are shown in the above figure.
The reward circuit (indicated in green) is a dopamine pathway from the ventral tegmental area to the nucleus accumbens (NAcc, also known as ventral striatum), which subsequently outflows through the ventral pallidum (VP). This pathway plays a role in responding to rewards and guiding behavior towards them. The encoding and reactivation of memories of drug use are regulated by the red areas. The hippocampus (HIP) is involved in place memories of drug use as well as the amygdala (Amyg) for the emotional aspects of these. Drug memories are known to be some of the most powerful aspects and they are extremely hard to overwrite or dislodge.
The purple areas of the anterior cingulate gyrus (ACG) and prefrontal cortex (PFC) are responsible for cognitive control and depend on a suitable balance of glutamate and GABA interactions, allowing this region to exert "top-down" control on the pleasure and motivation areas. The orbito-frontal cortex (OFC) evaluates the value of rewards or salience and the decision to use. Generally, these areas are in balanced condition, with the OFC having the final "say" in behavioral control, that is, "NO-GO" for drug use. This balance is disrupted in addiction, and the motivation to use drugs become stronger, drug memories become more dominant, and the "top-down" mechanisms become weaker such that drug use cannot be controlled, leading to a "GO" take drugs state.
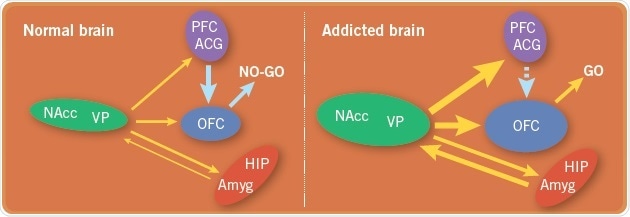
Fig 2. A comparison of the brain circuits in a normal brain with the brain circuits in an addicted brain. Image credit: Tocris Bioscience
Something must be different in the addict’s brain as not everyone who uses drug becomes addicted (Figure 2). This may be due to the reason for drug use in the first place or some adaptive changes induced by the drugs. Yet, recreational use of drugs also results in adaptive changes, for example, tolerance, that promotes continued use of drugs, because the withdrawal symptoms that emerge after the discontinuance of drugs is so severe that it drives relapse. With regards to alcohol, the κ-receptor-acting opioid peptide dynorphin is believed to be responsible for withdrawal symptoms. Ketamine, stimulants, and potentially opioids seem to decrease the function of the PFC/OFC, and therefore, they are less capable of exerting "top-down" control over urges and impulses.
Classes of Addictive Drugs
Stimulants are drugs that function by releasing dopamine and, in some instances, blocking its reuptake. This class contains amphetamine and its derivatives, for example, methylphenidate and methamphetamine (crystal meth), cocaine (and crack), and some cathinones, for instance, mephedrone. Their actions are based on the speed of brain penetration, with IV injection and smoking being the most addictive routes, but are highly unaffected by dopamine receptor antagonists. Other effects of neurotransmitters, such as endorphin release, could then describe their addictive properties.
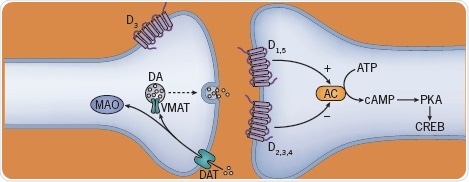
Fig 3. The effect of stimulants on neurotransmitters. Image credit: Tocris Bioscience
Drugs that imitate the actions of the endogenous opioid endorphin peptides are called opioids. They comprise of morphine, codeine, heroin, fentanyl, and methadone. They differ with respect to efficacy and affinity at the µ opioid receptor, which happens to be their site of action in reward. They are strong analgesics yet can lead to lethal respiratory depression. Buprenorphine, an example of partial agonists, are safer. These actions are blocked by antagonists such as naltrexone, nalmefene, and naloxone.
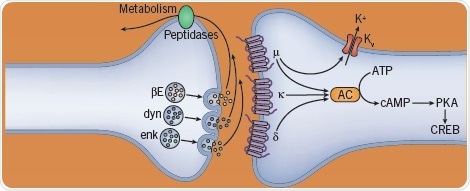
Fig 4. The effect of opioids on neurotransmitters. Image credit: Tocris Bioscience
Alcohol and other sedatives function mainly through the GABA receptor. Alcohol appears to improve the actions of both GABAB and GABAA receptors and at higher doses also blocks NMDA glutamate receptors, influences dopamine release, and stimulates 5-HT3 5-HT3receptors. Benzodiazepines are selective positive allosteric modulators (PAMs) of the α1, 2, 3, and 5 subunits of the GABAA receptor. Although a GABAB agonist, GHB also acts on GHB receptors. Barbiturates are GABAA PAMs, which at anesthetic doses also open the GABAA chloride ionophore directly.
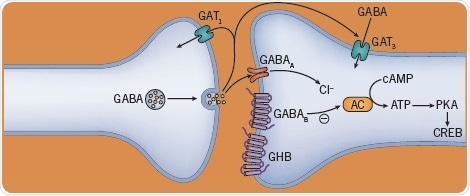
Fig 5. The effect of alcohol and sedatives on neurotransmitters. Image credit: Tocris Bioscience
Psychoactive cannabinoids, specifically ∆9 THC (the “stoning” element of herbal cannabis), imitate the effects of endogenous cannabinoids, for example, diacylglycerol and anandamide that are formed by the breakdown of phospholipids in cell membranes. The brain effects of cannabinoids are regulated through CB1 receptors, which are found at high concentrations in brain areas related to reward and motivation. They also act on CB2 receptors that are mostly found in the immune system. Another cannabinoid—cannabidiol — acts in some ways that oppose those of THC through mechanisms that are yet to be understood.
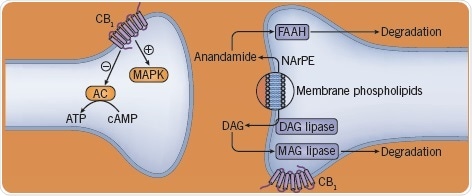
Fig 6. The effect of cannabinoids on neurotransmitters. Image credit: Tocris Bioscience
Ketamine, and previously PCP, are dissociative respiratory-sparing anesthetics that also treat chronic pain and acute depression. Conversely, they are addictive and can cause psychosis. They function as glutamate NMDA receptor antagonists but since several of these receptors are present presynaptically on GABA interneurons and on glutamate neurons, the ketamine blockade results in increased glutamate transmission, which, in turn, stimulates AMPA receptors. This consecutively can increase dopamine, which may be reinforcing.
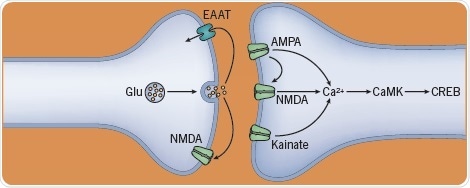
Fig 7. The effect of ketamine and PCP on neurotransmitters. Image credit: Tocris Bioscience
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.