Myocardial infarction (MI) – more regularly known as a heart attack – is an acute phenomenon initiated by the interruption of blood supply to regions of the heart, causing myocardial necrosis. Infarction of a significant area of the myocardium can inhibit regular heart conductance, engendering cardiac arrest. MI immediately follows the occurrence of an occlusive thrombus in a coronary artery, which blocks blood flow to the downstream tissue (Figure 1).
The most regular source of an occlusive thrombus in a coronary artery is the rupturing of an atherosclerotic plaque. Nonetheless, the occlusion of a coronary artery can also be caused by a coronary embolism. This can happen among patients who have undergone angioplasty, coronary artery bypass grafting, and stent placement. One of the few warning symptoms for MI is the presence of angina pectoris – an acute, cardiac-induced chest pain which can also radiate along the left arm.
Causes of Angina
Angina is caused by a deficiency of oxygen to the myocardium as a result of coronary artery obstruction or spasm and is classifiable as either ‘stable’ or ‘unstable’ angina. Patients with stable angina undergo ‘predictable’ chest pain during exertion, which is subsequently resolved after rest or the administration of the NO donor, nitroglycerin. The myocardium is only moderately damaged during stable angina. Conversely, an episode of unstable angina, which corresponds to chest pain occurring at rest or among patients with no history of stable angina, can lead to myocardial necrosis, albeit at a level lower than that which is observed during acute MI.
Two components of cardiac muscle whose serum levels rise as a result of myocardial necrosis, cardiac troponins T (cTnT) and I (cTnI), are the most typical blood biomarkers utilized in the diagnosis of acute MI or unstable angina. Although immediate pharmacological therapy of angina is accomplished by administering nitroglycerin, longer-term treatment requires either reducing metabolic demand of the heart by decreasing heart rate, through the administration of β-blockers or ivabradine, or increasing blood supply to the heart through the use of vasodilators, including calcium channel blockers, long-acting nitrates, and nicorandil.
Additional Pharmacological Procedure for Prevention of Angina
An additional pharmacological procedure for the prevention of angina is to increase ATP generation while maintaining identical oxygen consumption. One of the first antianginal agents, perhexiline, exerts these effects through the inhibition of the mitochondrial enzyme, carnitine palmitoyltransferase-1 (CPT1). Newer antianginal agents, comprising etomoxir and ranolazine, also modify fatty acid oxidation, augmenting metabolic efficiency, and preventing the occurrence of angina.
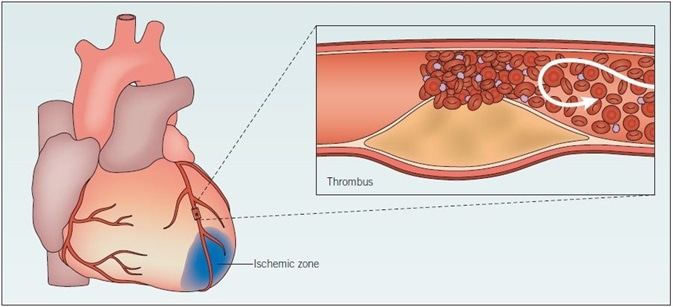
Figure 1. Myocardial infarction. When an atherosclerotic plaque ruptures, blood flow is greatly impeded, and thrombosis may occur. Consequently, the artery is occluded and the supply of oxygen to the heart is restricted. Ischemia results, and if it is sustained the heart muscle tissue may become damaged and die. The likelihood of plaque rupture is influenced by several factors, including elevated blood pressure and degree of inflammation. This damaged tissue does not function fully; acute myocardial ischemia results in ionic and metabolic disturbances that affect the membrane and action potentials of myocytes. Arrhythmias and heart failure may occur as a result of the slower conduction of electrical impulses.
As well as vasodilators, pharmacological therapy of severe MI also implements antiplatelet and fibrinolytic agents, such as recombinant human tPA, uPA, and streptokinase in conjunction with heparin, as a means of breaking down the occluding thrombus and restoring blood supply to the downstream myocardium. However, because it carries a lower risk of reocclusion and stroke, angioplasty is the preferred methodology for restoring vessel patency. The reduction of cardiac output by targeting the RAAS is also an effective therapeutic approach following MI, particularly among patients at risk of developing heart failure.
Adenosine receptor activation is also a useful therapeutic approach for reducing myocardial injury after MI. All four adenosine receptor subtypes – A1, A2A, A2B, and A3 – have been proven to be cardioprotective in the ischemic heart. Research is still identifying new targets for MI therapy. One of these targets is the cathepsin family, a category of enzymes that degrade myofibrillar proteins during MI and promote ventricular remodeling. The advantages of cathepsin inhibition could be two-fold – as well as lowering ventricular remodeling after MI, cathepsin inhibition might also inhibit atherosclerotic plaque rupture, the primary trigger of MI.
Additional targets
PI 3-kinase is an additional target in reducing ventricular remodeling and preventing MI-induced heart failure, although its ubiquitous expression restricts its usage as a therapeutic target. A more contemporary strategy for the treatment of myocardial infarction is based on the usage of stem cells in repairing damaged myocardium. After an infarction, damaged myocardium is unable to regenerate, and is thus replaced by non-contractile scar tissue. This modifies both the conductance and contractility of the myocardium and might cause the subsequent development of an arrhythmia or heart failure.
The injection of multipotent cardiac stem cells to the infarcted area of the heart, after myocardial infarction, has displayed potential in enabling regeneration of damaged myocardium, but their availability is restricted. Consequently, research endeavors are contemporarily focusing on generating cardiomyocytes via the differentiation of more readily available stem cell populations, comprising bone marrow-derived adult stem cells or undifferentiated skeletal myoblasts.
The induction of cardiomyogenic function in these stem cell populations has been accomplished utilizing an array of techniques. These comprise cardiac preconditioning, with which stem cells undergo differentiation in media previously utilized for culturing primary cardiomyocytes; and also the use of small-molecule inhibitors, including cardiogenol C and XAV 939 to regulate stem cell signaling pathways such as the Wnt/β-catenin pathway.
Enabling the repair and regeneration of damaged myocardium after infarction, in conjunction with the prevention of aberrant remodeling, constitute encouraging future therapeutic directions in the field of myocardial infarction studies.
About Tocris Bioscience
Tocris Bioscience is your trusted supplier of high-performance life science reagents, including receptor agonists & antagonists, enzyme inhibitors, ion channel modulators, fluorescent probes & dyes, and compound libraries. Our catalog consists of over 4,500 research tools, covering over 400 protein targets enabling you to investigate and modulate the activity of numerous signaling pathways and physiological processes.
We have been working with scientists for over 30 years to provide the life science community with research standards, as well as novel and innovative research tools. We understand the need for researchers to trust their research reagents, which is why we are committed to supplying our customers with the highest quality products available, so you can publish with confidence.
Tocris is part of the protein sciences division of Bio-Techne, which also includes the best in class brands R&D Systems, Novus Biologicals, ProteinSimple, and Advanced Cell Diagnostics. Bio-Techne has united these brands to provide researchers with a full portfolio of research reagents, assays, and protein platforms. For more information on Bio-Techne and its brands, please visit bio-techne.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.