Monoclonal antibodies (mAbs) have become the most growth-accelerated segment of the biopharmaceutical industry, with advanced molecular engineering and antibody humanization making it possible to create therapeutic antibodies for a host of medical conditions such as cancer, autoimmune disease, cardiovascular disorders and infectious disease.
These candidates are initially generated using a hybridoma technology or primary B cell screening following antigen immunization. In the process of antibody discovery, primary screens are important in identifying cell lines which possess essential attributes such as specificity of binding, cross-reactivity across species, selectivity and affinity.
Clones which satisfy these characteristics are assessed next for other vital properties, which may include IgG isotyping, antibody quantification, and cell count/health, which are necessary to select lead molecules, as shown in Figure 1.
Mouse antibody is quantified from cell supernatant using enzyme-linked immunosorbent assay (ELISA), a conventional technology which is nonetheless protracted and measures only one end point, as well as often needing sample dilution and multiple wash steps.
In addition to this, other tests must be carried out such as separate IgG isotyping and cell number/health, before sufficient insight is available for downstream cloning of the antibody. This traditional workflow can be streamlined using the Sartorius Mouse IgG Type and Titer Kit.
This is a high throughput multiplexed assay which has a wide dynamic range, which avoids sample dilution and wash steps. It offers a simple workflow with the capability to simultaneously measure five different endpoints:
- IgG isotype
- IgG quantity per isotype
- Total IgG secretion level
- Cell count
- Cell health
The high-content data enables rapid differentiation between monoclonal and polyclonal wells. It tells the researcher how much IgG is produced, for quantifying stable clones and for downstream functional assays.
It identifies the isotype to help design the right primer for PCR-mediated gene cloning. Finally, it monitors the proliferation and health of cells before the isolation of RNA is performed.
The Mouse IgG Type and Titer assay is one component of Sartorius' integrated antibody discovery platform. Another solution is the iQue3 PLUS platform which can speed up data acquisition, and miniaturize assays to a 384-well plate. After a 20-minute data acquisition, ForeCyt software provides powerful data mining tools to enable large-scale hybridoma/B-cell screening studies.
The Panorama feature on ForeCyt Software provides dynamic multi-plate data analysis, including profile maps with user defined criteria to detect and identify those samples that contain the required IgG isotype antibody concentration and cell health, which are very important when analyzing large, multi-plate screening campaigns. In short, the Mouse IgG Type and Titer assay is capable of simplifying and accelerating the workflow in antibody discovery, which means less time to achieve actionable results.
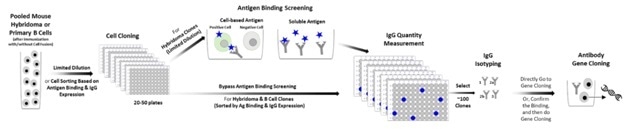
Figure 1. Traditional Antibody Discovery Workflow for Lead Candidate Generation. After antigen immunization, pooled mouse hybridoma cells (post fusion) or primary mouse B cells are grown in microtiter plates using limited dilution or single cell sorting. Primary screening of the cell clones is performed to identify clones with the desired antigen binding characteristics. Positive clones are verified for mouse IgG secretion. Using antigen binding cell sorting methods single cell clones go directly to the IgG quantification step. Typically, the 100 top positive clones are characterized by IgG isotyping. These samples will be used directly for gene cloning or sequencing. Alternatively, the positive clones are tested in functional assays after normalization of IgG content. After further selection by functionality, the selected cell clones will be used to construct antibody expression vectors.
Materials and Methods
Samples and Reagents
This experiment describes mouse hybridoma studies using a single plate of 72 hybridoma clones. These were first evaluated, alongside a 12-point standard IgG curve in duplicate. The samples which had cell supernatant, either alone or in mixture with cells, were then analyzed with the Mouse IgG Type and Titer Assay as per protocol.
B-cell culture assay performance was measured using the supernatants from about 450 single cell mouse primary B-cell cultures, after transferring them to five plates of 96 wells each. A separate plate was used to generate IgG standard curves.
Hybridomas typically produce higher antibody levels than B-cell samples, which was compensated for by using the alternative high-sensitivity assay described in the protocol of this kit.
Data Acquisition and Analysis Workflow
The process of analysis was streamlined by the inclusion of a USB drive which has a template for data acquisition and analysis. This means the platform is essentially plug-and-play, containing the required plate design, the protocol for sampling, the gates for the cells and beads, and heat maps.
When data is acquired, the events are populated within the predefined gates in the histograms and dot plots in real time. On completion of data acquisition, the ForeCyt software automatically calculates the heat maps and resulting metrics for each data point.
The standard curves for isotype specific IgG and total IgG may be calculated either from the assay plate or from another plate. In a screening campaign one standard curve is sufficient for all plates if the Panorama tool is used, since it allows multiple plate visualization.
The assay template enabled identification of monoclonal hits as specified by a single IgG isotype and the quantity of IgGs in each sample was also quantified. For multi-plate analysis, profile maps were created with multi-parameter, user defined criteria with the Panorama feature in the ForeCyt software. Multi-plate line graphs and criteria rankings were rendered using Panorama.
Results and Discussion
Mouse IgG Type and Titer Workflow
As illustrated in Figure 2, the multiplex assay has a streamlined workflow, which allows the experiment to be set up and data to be acquired more rapidly, unlike conventional ELISA which needs more steps in the sample preparation and processing.
The lower dynamic range of ELISA is the reason why multiple dilutions of the supernatants of hybridoma cultures need to be made, so that the samples remain within the linear range of the test. However, the Mouse IgG Type and Titer assay has a wider dynamic range of 0.05 µg/mL to 50 µg/mL, which allows determinations within the range of concentrations that are typical of hybridomas and B-cell antibody production.
The samples can therefore be directly placed in the assay plates, whether cell-based or not, without having to be either centrifuged to remove the cells or diluted with much labor. Every reagent that is required is added to the sample directly, without the need for washing steps.
This is termed the mix-and-read format, resulting in less set-up time as well as improved reproducibility. From sample transfer and reagent preparation onwards, a 96-well plate can be set up in about 15 minutes, and even for a 384-well plate the time required is only a little longer.
The time for incubation is one hour. Sample acquisition takes from 6 to 20 minutes depending on whether the 96 or 384-well format is used. The data acquisition template is then brought into play, keeping the whole processing time from samples to the output of actionable data under 100 minutes.
Mouse IgG Type and Titer Biochemistry
The Mouse IgG Type and Titer kit operates as a multiplexed competition assay(Figure 2). The assay uses four types of capture beads, each one coated with antibodies directed against one mouse IgG isotype.
The detection reagent is a mixture composed of four separate mouse IgGs labeled with FITC. In each sample well, the IgG isotypes in the supernatant compete with the labeled IgG corresponding to each one for binding to their specific beads.
The IgG isotype concentration will therefore be inversely related to the isotype-specific capture beads which are detected by their fluorescence. An isotype-specific standard curve is generated using which the concentration of the sample IgG isotype can be found. If samples contain both culture supernatant and cells, cell count and viability parameters are also measured.
The Mouse IgG Type and Titer assays have a number of aspects of superiority to conventional assays:
- IgG isotype identification: The assay determines the different IgG isotypes from a single clone, thereby detecting polyclonal cultures. This helps to rule out these wells and concentrate upon the monoclonal wells, reducing the number of subclone cultures that need to be made and shortening the development of the mAb. Another advantage is the use of isotype-specific DNA primer sets once the isotype is known, rather than having to use degenerate primers to clone or sequence variable regions in the antibody encoding region.
- Precise total and isotype specific IgG quantification: Measurement of the quantity of IgG helps normalize the concentration of IgG for all clones. This is essential before any confirmatory or functional assays are carried out downstream. The capture beads are also multiplexed to one of four specific IgG isotypes, and this means each of these isotypes can be quantified precisely. Conversely, ELISA and other singleplex assays have a single “total” IgG antibody, to which the binding affinity of the various IgG isotypes may be quite different. This means a generic rather than specific standard curve is drawn, and therefore each of the isotypes in the sample cannot be accurately measured using it. The Mouse IgG Type and Titer assay is different in that it makes use of capture beads which are coated with four different antibodies (IgG1, IgG2a, IgG2b, IgG3). This allows the generation of four different standard curves, one for each isotype, and accurate quantitative determination of each of the isotypes in the sample.
- The cell count and cell health are measured at the same time: Cell health as well as cell proliferation are key to the selection of cells for RNA extraction and subsequent PCR-dependent antibody cloning.
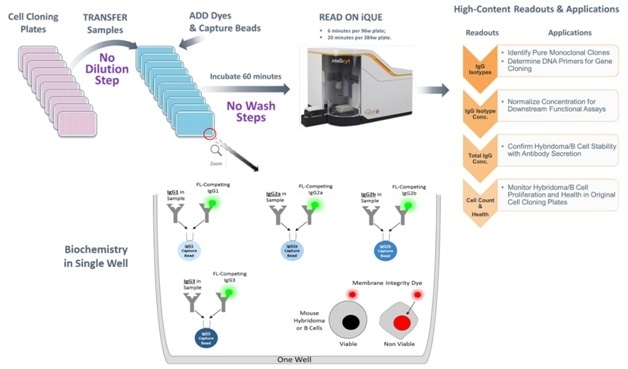
Figure 2. Mouse IgG Type and Titer Workflow and Assay Biochemistry Samples from hybridoma or B-cell cloning plates are directly transferred to the assay plate without an intermediate dilution step. Each assay well is seeded with four different types of capture beads, each with a specific affinity for a single IgG isotype. The IgG isotype in the sample will compete with the same FITC-labeled IgG isotype to bind to the capture bead. The IgG isotype quantity will be calculated from the isotype-specific standard curve. If cells are included in the assay well, a membrane integrity dye in the detection reagent will stain the membrane-compromised dead cells by DNA intercalation. After the addition of the detection reagent mixture and the capture beads to the samples and 60-minute incubation, the samples are acquired on the iQue3 PLUS.
Data analysis of a 96-well Hybridoma Plate Using the Mouse IgG Type and Titer Kit
When a high-content assay is run, the analysis of resulting data takes time and many complexities are involved. The mouse IgG Type and Titer kit takes full advantage of the analytical algorithms used by the Sartorius ForeCyt software’s system to identify hits quickly.
In the present experiment a 96-well plate containing mouse hybridoma cells was used. Limited dilutions were used to prepare the cell samples. Two weeks later, 20 µL of sample (cells and supernatant) was removed from 72 clones and placed in an assay plate for incubation after adding the reagents.
After this, samples were acquired on the iQue3 PLUS as shown in Figure 3. The template was then used to automatically produce four standard curves for the four isotypes, with the sample IgG isotypes being quantified and populated in heat maps and a data grid.
The same format was used to automatically populate cell number and cell health data. Finally, Panorama was used to visualize all the isotypes in the sample wells by generating an overlay line graph displaying all the isotypes and the quantity of each in all the samples.
This enables plate-level monitoring and quality control assessment of screening results. The whole process from acquiring data to identifying molecules which could be candidates for further research takes only a few minutes for each plate.
Data Analysis of a Multi-Plate B-Cell Screening Campaign
This experiment involved the analysis of 20 µL of supernatant from five 96-well plates containing primary B-cell clones, which was performed with the Mouse IgG Type and Titer kit’s high-sensitivity protocol, due to the relatively smaller amount of antibody secreted by B cell cultures. The process is shown in Figure 4.
The standard curves specific for each isotype were created from another standard plate, and the ForeCyt software generated automatic calculations of the IgG amount in each sample from these curves.
The Panorama feature populated this data in the data grid in the five plates. User-defined criteria were also used, namely, wells that had above 0.1 ug/ml IgG1 and below 0.1 ug/ml of all other isotypes, to create profile maps which showed specific hits for all the plates at screening level.
Any sample fulfilling these criteria was ranked on a line graph created by Panorama on the basis of IgG1 concentration. From data acquisition analysis onwards, the screening campaign was completed in about an hour.
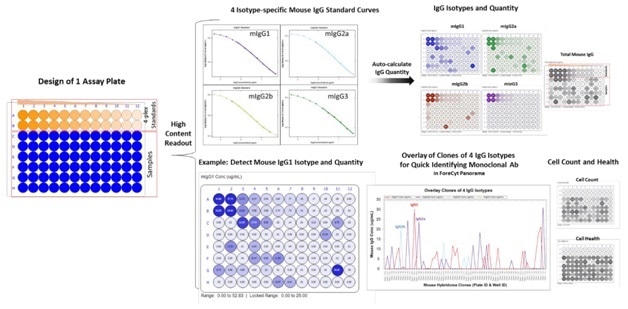
Figure 3. Analysis of High-content Readouts from Hybridoma Samples using Mouse IgG Type and Titer Kit. A single microtiter plate was designed with in-plate 4-plex standards for the top two rows and with hybridoma cell/supernatant samples in the bottom six rows. After data acquisition into the assay template, the analysis algorithm automatically generates four isotype-specific standard curves and IgG concentration is quickly calculated for each sample. The analysis software also provides the IgG isotyping and clone purity information in an overlay line graph in the Panorama function of ForeCyt. Cell count and health/viability is displayed in heat maps or in a data grid.
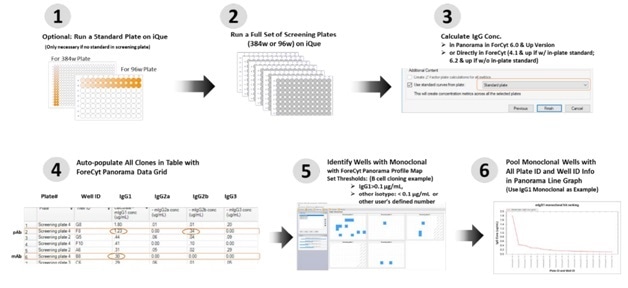
Figure 4. Data Acquisition and Analysis Workflow in a B-cell Screening Campaign using the Mouse IgG Type and Titer Kit Supernatants from ~ 450 clones were analyzed in a multi-plate screening study. IgG quantitation at the screening campaign level, and profile maps were generated to identify monoclonal samples containing high levels of IgG1. Samples meeting the user defined criteria were ranked by IgG1 concentration using a line graph.
Conclusion
The Mouse IgG Type and Titer kit is a novel assay platform for the discovery of mouse antibody, which succeeds by simplifying conventional assay workflows and combining three assays which used to be performed separately into one high throughput multiplex assay.
It uses the competition binding to enable a wide dynamic range for the detection of IgG, thus preventing the need to dilute and wash the samples, and reducing possible sources of variability.
The multiplex nature of the test means that high content data is available for analysis, to analyze specific antibody cell clone parameters and pick out samples with increased monoclonal IgG secretion, with high cell health, while excluding the polyclonal samples, those with low antibody production and those with unhealthy cells.
The visualization tools in ForeCyt software including Panorama algorithm for analyzing multiple plates gives researchers the broader picture so that candidate antibody molecules can be rapidly screened.
The Sartorius Mouse IgG Type and Titer assay is an innovative platform which can be combined with powerful software for data analysis to markedly shorten the time taken to analyze the data and reach sound decisions on its basis.
Acknowledgements
Produced from materials originally authored by Zhaoping Liu and John O’Rourke, Sartorius Albuquerque, New Mexico.
 Sartorius
Sartorius
Sartorius is a leading international pharmaceutical and laboratory equipment supplier. With our innovative products and services, we are helping our customers across the entire globe to implement their complex and quality-critical biomanufacturing and laboratory processes reliably and economically.
The Group companies are united under the roof of Sartorius AG, which is listed on the Frankfurt Stock Exchange and holds the majority stake in Sartorius Stedim Biotech S.A. Quoted on the Paris Stock Exchange, this subgroup is comprised mainly of the Bioprocess Solutions Division.
Innovative Technologies Enable Medical Progress
A growing number of medications are biopharmaceuticals. These are produced using living cells in complex, lengthy and expensive procedures. The Bioprocess Solutions Division provides the essential products and technologies to accomplish this.
In fact, Sartorius has been pioneering and setting the standards for single-use products that are currently used throughout all biopharmaceutical manufacturing processes.
Making Lab Life Easier
Lab work is complex and demanding: Despite repetitive analytical routines, lab staff must perform each step in a highly concentrated and careful way for accurate results.
The Lab Products and Services Division helps lab personnel excel because its products, such as laboratory balances, pipettes and lab consumables, minimize human error, simplify workflows and reduce physical workloads
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.