Radioligand binding experiments in pituitary tumor cell lines revealed that somatostatin (SST) may efficiently suppress the release of prolactin and GH (growth hormone), which led to the discovery of SSTR2. SSTR2 is broadly distributed in numerous tissues, according to subsequent radioligand binding tests.
SSTR1-SSTR5 are the five distinct SSTR genes discovered, with SSTR2 being the most investigated.
The expression of SSTR2
In normal tissues, SSTR2 mRNA and protein are broadly expressed; however, their expression is inconsistent. SSTR2 protein has the greatest levels of expression in the brain and kidneys. SSTR2 is extensively expressed in solid tumors. SSTR2 is now the most important target for the therapy of neuroendocrine tumors (NETs) due to its high expression in these cancers.
SSTR2 is shown to be highly methylated in colorectal cancer and has also been linked to carcinogenesis in gastric and breast cancers. SST binding to SSTR2 also prevented the production of cytokines from immune cells, which affected the tumor microenvironment (TME), according to the researchers.
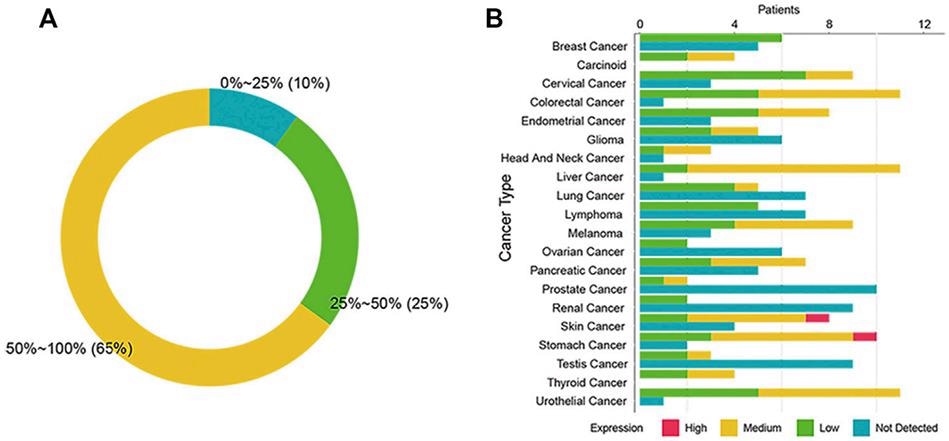
The Expression of SSTR2 in 20 cancers from the Human Protein Atlas. Image Credit: ACROBiosystems
The researchers from Xinqiao Hospital of Third Military Medical University used IHC data from the Human Protein Atlas to investigate the expression of SSTR2 in various cancers.
The findings revealed that the IHC positive rate of SSTR2 in 13 tumors was greater than 50%; 5 cancers somewhat expressed SSTR2 (positive rate 25–50%), and 2 cancers slightly expressed SSTR2 (positive rate 0–24%).
It can be shown that SSTR2 expression varies and is extensively spread across distinct cancer types. Furthermore, in patients with high SSTR2 expression, cell-mediated immunity may be better triggered and sustained.
The structure and function of SSTR2
SSTR2 is a seven-pass transmembrane protein also with N-terminal extracellular and the C-terminal intracellular. It belongs to the G protein-coupled receptor (GPCR) family. Some SSTRs have been reported to heterodimerize with other SSTs and other GPCRs, such as DRD2, β-adrenergic receptors, in studies.
SSTR transport, signal transduction, and biological activity can all be affected by the co-expression of these interacting receptors in cells. As a result, further study into the function of these heterodimers in physiological or pathological situations is needed.
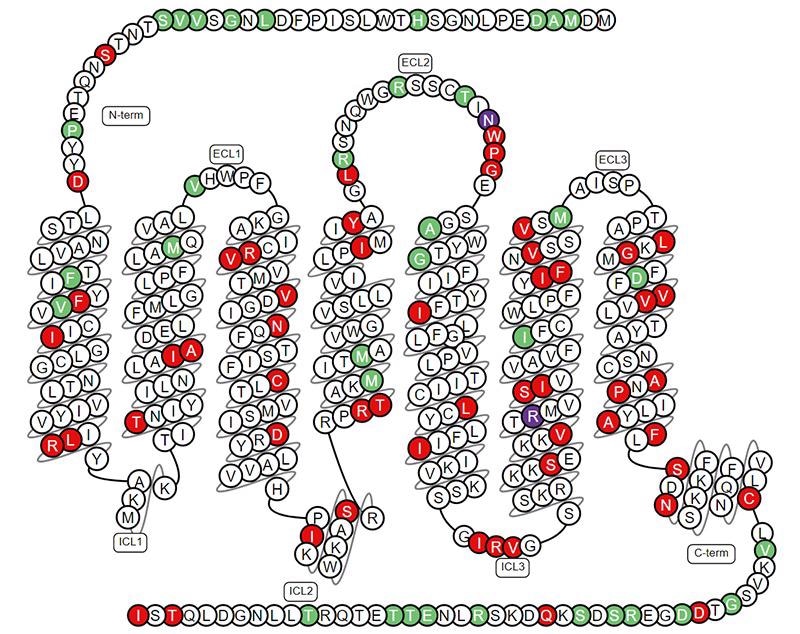
The Structure of SSTR2. Image Credit: ACROBiosystems
By binding to the SST, SSTR2 prevents the release of several hormones and other secreted proteins, reducing cell proliferation as well as angiogenesis. SST has anticancer action via both direct and indirect mechanisms:
- Binding to SSTR on the tumor surface triggers cell cycle arrest or apoptotic mechanisms downstream.
- SSTR causes tumor angiogenesis and tumor growth inhibitor production. In 90% of human pancreatic tumors, the SSTR2 gene is not expressed. The expression of its ligand (SST) is induced in human pancreatic cells that express SSTR2 (without expressing endogenous SSTR2), reducing cell proliferation, tumorigenicity, and metastasis.
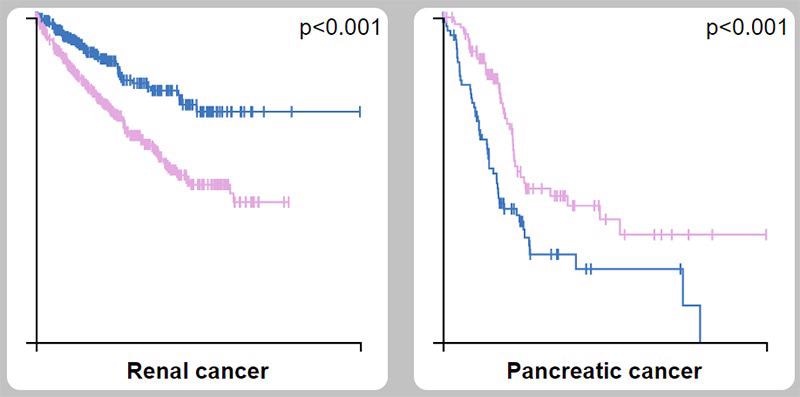
SSTR2 as a prognostic marker in kidney cancer (unfavorable) and pancreatic cancer (favorable). Image Credit: ACROBiosystems
Representative clinical therapeutic drugs targeting SSTR2
Xencor has developed Tidutamab (XmAb-18087), a representative drug that targets SSTR2. XmAb-18087 is a bispecific antibody (IgG1) that binds to CD3 to activate T cells, allowing them to destroy SSTR2-expressing tumor cells efficiently and precisely. Neuroendocrine tumors and lung cancer are the major indications for XmAb-18087, which is now in Phase I/II trials.
ACROBiosystems has successfully produced full-length SSTR2-VLP protein (Met 1-Ile 369) in HEK293 cells using “FLAG” to aid drug and therapy development targeting SSTR2.
Product list
Source: ACROBiosystems
| Molecule |
Cat. No. |
Host |
Product Description |
Application |
| SSTR2 |
SS2-H5216 |
HEK293 |
Human SSTR2 Full Length Protein-VLP (HEK293) |
Immunization
ELISA
SPR/BLI
Cell-based Assay
… |
| VLP |
VLP-N5213 |
|
Virus-Like Particle (VLP) isotype control VLP |
Isotype Control |
SSTR2: Product features
- Full-length SSTR2 protein with native and complete conformation
- Due to VLP’s intrinsic properties, it has a higher immunogenicity
- Binding to antibodies confirms high biological activity
- Dendritic cells and phage display can employ targets that are 100–300 nm in size
- Immunization/ELISA/SPR/BLI/cell-based assays, and so forth
SSTR2: Verification data
Full-length SSTR2 6-VLP protein (Cat. No. SS2-H5216)
High bioactivity verified by ELISA
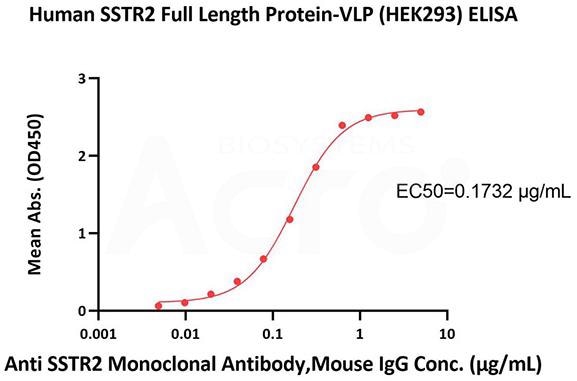
Immobilized Human SSTR2 Full Length Protein-VLP (Cat. No. SS2-H5216) at 10 μg/mL (100 μL/well) can bind Anti SSTR2 Monoclonal Antibody, Mouse IgG with a linear range of 0.005-0.625 μg/mL (QC tested). Image Credit: ACROBiosystems
Reference
- Marily Theodoropoulou, Günter K. Stalla. Somatostatin receptors: From signaling to clinical practice. Frontiers in Neuroendocrinology (2013). http://dx.doi.org/10.1016/j.yfrne.2013.07.005
- Aoyun Wang, Yixiao Yuan, Han Chu, et al. Somatostatin Receptor 2: A Potential Predictive Biomarker for Immune Checkpoint Inhibitor Treatment. Pathol. Oncol. (2022) https://doi.org/10.3389/pore.2022.1610196
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.