
Image Credit: ACROBiosystems
Recent advancements in organoid structures have allowed scientists to culture complex collections of cells that mimic the architecture and functionality of a patient’s organs, imitating the environment of their cellular tissue and organs.
These three-dimensional organoid structures contain populations of self-renewing stem cells, which can separate into distinct cell types in the organ tissues.
Organoids have richer compositions and improved physiological functions compared to traditional two-dimensional models and thus have widespread use in modeling disease, drug screening, biological function research, and the development of artificial organs.
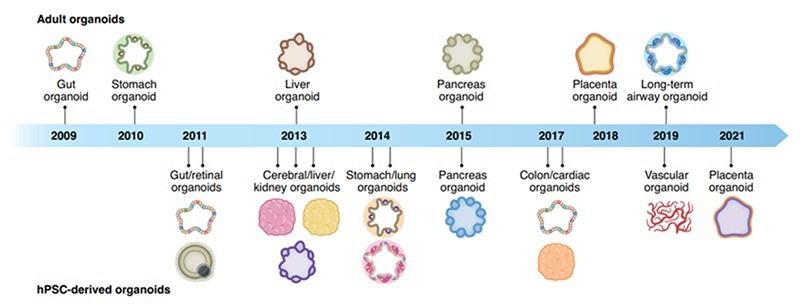
Figure 1. Organoid discovery of their corresponding organs through the years.1 Image Credit: ACROBiosystems
Organoid sourcing and culturing: Stem cells and growth factors
Organoids can be split into adult stem cells (ASCs) and pluripotent stem cells (PSCs). PSCs further comprise embryonic stem cells and induced pluripotent stem cells.
ASCs maintain stem cell potential in adult human organs, which can subsequently conserve homeostasis or damage the repair processes of organs. They are organ-specific, and ASC organoids accurately represent adult tissues' physical and practical characteristics.
PSCs alternatively must undergo various differentiation processes, simulating the entire organ development process, and are, therefore, a vital tool for studying organ development and gene function.
Both stem cell sources can mimic most major organs and tissues, but ASCs are more widely used. However, there are exceptions where tissue is difficult to obtain (for instance, brain tissue); in these circumstances, PSCs are considerably superior.
Despite differences between stem cell sources, growth factors play crucial roles in inducing and influencing the differentiation of cells. Organoid research requires enhanced extracellular matrix compositions to guarantee good physiological development; organoid culturing often contains several growth factors and can vary significantly between types of organoids.
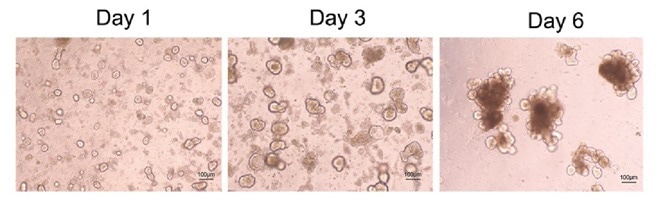
Figure 2. Confocal microscopy of mouse intestinal organoid growth with growth factors: human EGF (Cat. EGF-H52H3), Noggin (Cat. NON-H5257), and R-spondin1 (Cat. RS6-H4220). Image Credit: ACROBiosystems
Application of organoids in research
There are three key areas of research in which organoids have a vital position: drug discovery, precision medicine, and regenerative medicine.
Organoids are perfect for high-throughput drug screening in precision medicine, where cultured organoids from patient biopsies are exposed to xenobiotics to screen for optimal effects, facilitating personalized medication and therapy.
Organoids are exceptional for drug screening in a range of tumor types, including colorectal, breast, non-small cell, and gastric cancer. Drug-response prediction by organoids in some tumors has been demonstrated to be accurate up to 80%, suggesting incredible potential in personalized diagnostic medicine.
Organoids represent a process which is, respectively, more accurate and cost-effective than traditional cell lines and animal models. The complex tissue structure, cell makeup, and physiological structure of organoids, alongside their high repeatability and throughput of use in in vitro studies, make organoids ideal for preclinical drug screening.
Gene editing can also be employed to build various models for diseases, including cystic fibrosis, microcephaly, and colorectal cancer. Organoids can be used to study the process of host–pathogen interactions in infections such as COVID-19 and Heliobacter pylori; scientists have constructed various organ models to study the infection process and how it influences changes in physiological function in the former.
The 20 largest pharmaceutical companies in the world (e.g., Novartis, Pfiizer, Johnson & Johnson) all use organoids to assess the safety and effectiveness of new medications.
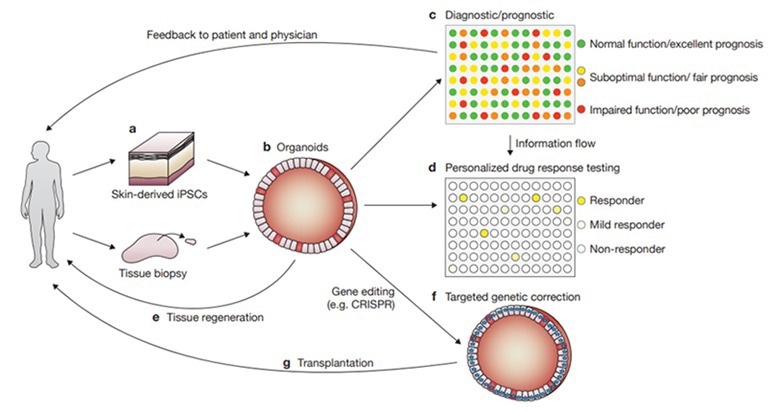
Figure 3. Various applications of organoids for disease research, drug development, and personalized medicine.2 Image Credit: ACROBiosystems
Organoids also test the potential of repairing damaged tissues in regenerative medicine. For instance, researchers have assessed the possibility of transplanting organoids to treat inflammatory bowel disease and short bowel syndrome. Scientists have also initiated attempts to treat type I diabetes using islet organoids, but these studies are in their first stages and are currently limited to animal models.
Organoids have drawbacks in clinical applications, including the heterogeneity of cultured organoids, the in vivo homing effect post-organoid transplantation and the potential tumorigenicity of matrix gels in organoid cultures, but their potential in regenerative medicine is undeniable.
The future of organoids
Academics are constantly advancing organoid cultivation and have recently discovered which growth factors are necessary to direct cell differentiation; organoids improve greatly on two-dimensional cell cultivation and have become indispensable in developing and evaluating medication. They can mimic human organs' physical and functional features and simulate the tumor microenvironment.
Organoids are also set to become essential preclinical models for disease prevention. Almost all human organs can be formed into organoids in vitro.
To support research on organoid cell culturing, ACROBiosystems has developed a series of high-quality cytokines, including EGF, Noggin, R-Spondin 1, FGF10, FGF2 and Activin A. These products are suitable as growth factors for organoid culture that have been verified to promote organoid growth.
References and further reading
- Han, Y., Chen, S., et al. (2022) Human Organoid Models to Study SARS-CoV-2 Infection. Nature Methods, 19, pp.418-428.
- Fatehullah, A., Barker, N., et al. (2016) Organoids as an In Vitro Model of Human Development and Disease. Nature Cell Biology, 18(3), pp.246-254.
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.