The COVID-19 pandemic continues to affect communities, families, and individuals all over the globe. The early testing of the disease will lead to fast identification of cases, fast treatment for those individuals and immediate isolation to stop the disease spreading.
In the bigger public health picture on mitigation efforts, the diagnosis of COVID-19 is also crucial as it helps investigators to characterize the spread, prevalence, and contagiousness of the disease.
At present there are two main types of tests for COVID-19, the molecular test and the serological test. Also known as the Nucleic Acid Amplification Tests (NAAT), the molecular test detects the genetic material from SARS-CoV-2, which causes COVID-19.
This technique is recommended by the FDA and WHO as the gold standard for COVID-19 diagnosis. Yet, the molecular test has its drawbacks. Detecting SARS-CoV-2 from pharyngeal swabs needs high-quality specimens that have enough intact viral RNA.
The virus loads differ significantly in the respiratory tract, which is further exposing healthcare workers to a risk of infection because it has led to high false-negative rates, with probable cases staying negative after numerous swabs.
The serological test detects the antibodies in the blood sample, unlike the molecular test. This is typically gathered through a finger prick, giving better patient compliance. Furthermore, the serological test is able to identify those who were infected and have recovered, which supplies a bigger picture of COVID-19 infection spread.
Usually, the serological test is quicker than the molecular test and does not need special equipment to process the results. Numerous countries around the world are planning to use this technique as an additional diagnostic tool because of the convenience and importance of the serological test.
For instance, Germany is performing nationwide testing for COVID-19 antibodies, becoming the first European country to do so.
The serological test can be utilized in different formats, including enzyme-linked immunosorbent assay, colloidal immuno-chromatographic strip, and chemical immunoluminescence techniques, while the principles behind each format are fairly similar. Take the chemical immunoluminescence technique for example.
The antigen immobilized beads are able to capture the specific antibodies from the patient sample (i.e. blood sera). After adding a secondary antibody reporter to the system, the virus-specific immune signal is produced to confirm the presence of a past or ongoing viral infection, as seen in Fig 1.
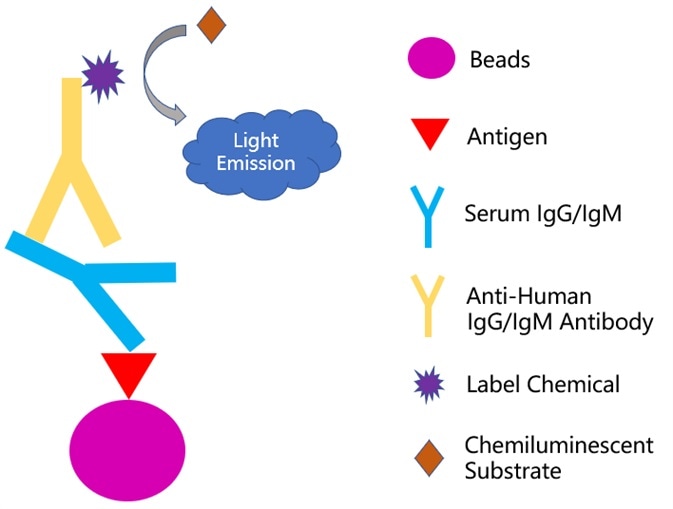
Figure 1. Principle of chemiluminescence-immunoassay method. Image Credit: ACROBiosystems
In the US so far, the FDA has authorized four serological test kits for emergency utilization, they are summarized in Table 1. There are many studies taking place to evaluate these test kits as the importance of the serological test is growing. Recently, work was shared online by a team from the Southern University of Science and Technology.[1]
Table 1. Antibody test kit manufacturers and commercial laboratories authorized for emergency use by FDA. Source: ACROBiosystems

By utilizing the chemiluminescence immunoassay technique, which was previously explained in detail (Fig.1), they developed a serological test for the diagnosis of SARS-Cov-2.
In order to capture the IgM and IgG induced by SARS-CoV-2, the recombinant nucleocapsid protein of SARS-CoV-2 was successfully expressed from E. coli. and then immobilized to the magnetic beads.
51 tuberculosis patients, 29 healthy individuals, and 79 SARS-CoV-2 patients were enrolled randomly in this study. In the IgM testing, the average RLU (relative light units) of the patient group was higher than the control groups which were made up of health cohort and tuberculosis patients.
The difference is even more significant when it comes to the IgG testing, as seen in Fig.2. A Receiver Operating Characteristic (ROC) curve was gathered based on all the RLUs values.
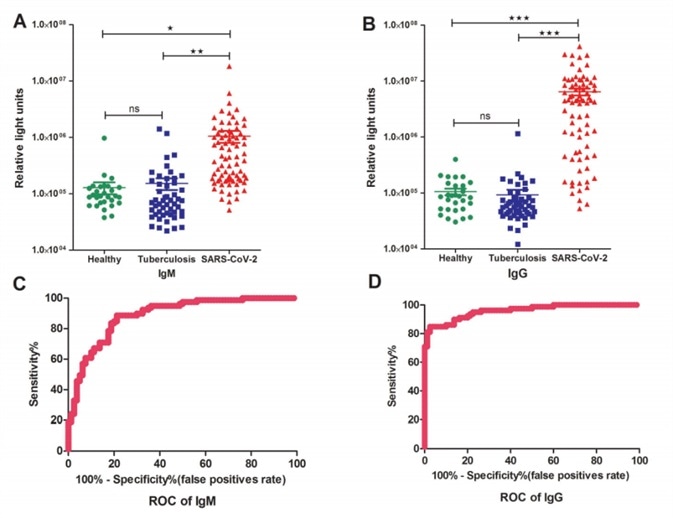
Figure 2. Detections and measurements of the SARS-CoV-2 IgM and IgG antibody in healthy people, tuberculosis patients and SARS-CoV-2 confirmed patients using the chemiluminescence immunoassay method (A and B). The average results were expressed as mean ± SEM of all individuals. Receiver Operating Characteristic curves for IgM (C) and IgG(D) were obtained based on the RLU for the SARS-CoV-2 patient group and the control group consisting of healthy cohorts and tuberculosis patients. Image Credit: Evaluations of serological test in the diagnosis of 2019 novel coronavirus (SARS-CoV-2) infections during the COVID-19 outbreak.
Both IgM and IgG testing have predictive values with a cutoff setting for IgM (RLU 162296) and IgG (RLU 336697), in which the calculated sensitivity and specificity for IgM were 82.28% (65/79) and 81.25% (15/80), and 82.28% (65/79) and 97.5% (2/80) for IgG, respectively, according to the ROC curve.
Despite the onset time or gender, no statistically significant difference was seen between IgM and IgG testing results. It was discovered that there was a significantly lower detection rate in both IgM and IgG testing for individual groups younger than 18 years old, which was probably associated with no or less production of antibodies in them.
These results were also compared with a commercially available ELISA kit and discovered that their chemiluminescence technique had significantly higher sensitivity. The sensitivity difference may be partially due to the different serum amounts for the incubation step. Although, the intrinsic technique difference can also contribute.
A well-developed serological test with high specificity and sensitivity is enough to become a diagnostic tool for COVID-19. The serological test should gain more attention as a vital supplement to the molecular test.
Nucleocapsid protein is a crucial part of SARS-CoV-2. The nucleocapsid protein is always selected to capture IgG/IgM in the COVID-19 serological kits because of its high immunogenicity.
As the vital component in the serological test kit, the recombinant nucleocapsid protein of SARS-CoV-2 has a huge influence on the specificity and sensitivity.
ACROBiosystems has developed different types of N protein. The binding activity of N protein with anti-N protein antibody was verified by ELISA, with a high sensitivity of 0.02 ng/mL.
Product: SARS-CoV-2 (COVID-19) Nucleocapsid protein, his tag (C-terminus)
Cat. No.: NUN-C5227
The sensitivity of N protein (His tag at the C-terminus) with anti-N protein antibody was 0.02 ng/mL as verified by ELISA.
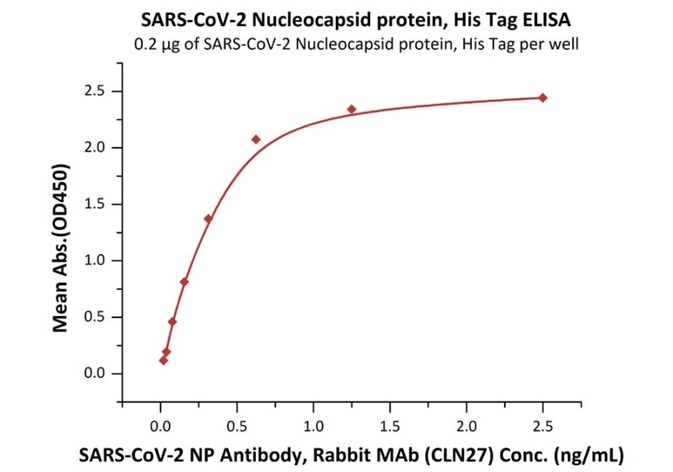
Immobilized SARS-CoV-2 Nucleocapsid protein, His Tag (Cat. No. NUN-C5227) at 2 μg/mL (100 μL/well) can bind SARS-CoV-2 NP Antibody, Rabbit MAb (CLN27) with a linear range of 0.02-0.6 ng/mL. Image Credit: ACROBiosystems
Product: SARS-CoV-2 (COVID-19) Nucleocapsid protein, his tag (N-terminus)
Cat. No.: NUN-C51H9
The sensitivity of N protein (His tag at the N-terminus) with anti-N protein antibody was 0.02 ng/mL as verified by ELISA.
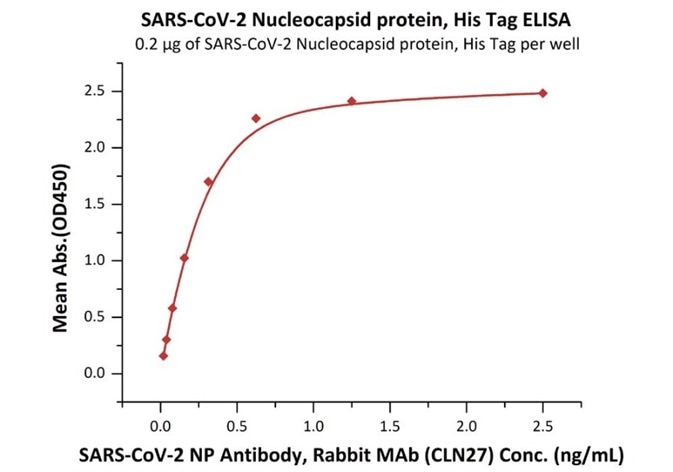
Immobilized SARS-CoV-2 Nucleocapsid protein, His Tag (Cat. No. NUN-C51H9) at 2 μg/mL (100 μL/well) can bind SARS-CoV-2 NP Antibody, Rabbit MAb (CLN27) with a linear range of 0.02-0.3 ng/mL. Image Credit: ACROBiosystems
Product: Biotinylated SARS-CoV-2 (COVID-19) Nucleocapsid protein, his,avitag™
Cat. No.: NUN-C81Q6
The sensitivity of biotinylated N protein with anti-N protein antibody was 0.02 ng/mL as verified by ELISA.
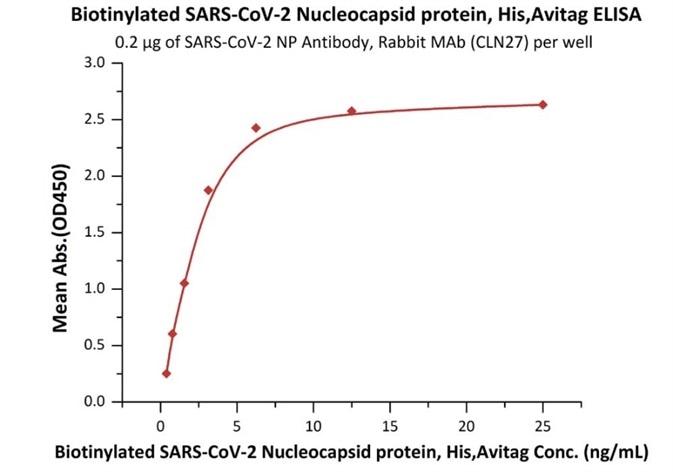
Immobilized SARS-CoV-2 NP Antibody, Rabbit MAb (CLN27) at 2 μg/mL (100 μL/well) can bind Biotinylated SARS-CoV-2 Nucleocapsid protein, His,Avitag (Cat. No. NUN-C81Q6) with a linear range of 0.4-3 ng/mL. Image Credit: ACROBiosystems
References
- Dachuan Lin et al. Evaluations of serological test in the diagnosis of 2019 novel coronavirus (SARS-CoV-2) infections during the COVID-19 outbreak. medRxiv preprint doi: https://doi.org/10.1101/2020.03.27.20045153
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.