Human immunodeficiency virus (HIV) infection is a viral infection that causes acquired immunodeficiency syndrome (AIDS) by gradually destroying certain leukocytes. When HIV enters the human body, it targets human immune cells (primarily CD4+ T lymphocytes).
The primary purpose of the human immune system is diminished and ultimately destroyed, owing to the huge death of immune cells.
AIDS patients will eventually be vulnerable to a variety of diseases, opportunistic infections, and malignancies. Despite the fact that HIV is a multifactorial disease, research has shown that specific targetingof CXCR4 and CCR5 is a potential therapeutic tactic for HIV as well as other immune situations like tumors.
Introduction to CXCR4
CXCR4 is a member of the G protein-coupled receptor family (GPCRs). CXCR4 is a seven-transmembrane protein with an overall length of 352 amino acids that forms a circuitous surface receptor with -NH2 on the outside and -COOH on the inside. CXCR4 is encoded by a gene on human chromosome 2q21 and is primarily demonstrated in monocytes, neutrophils, lymphocytes, and activated T cells.
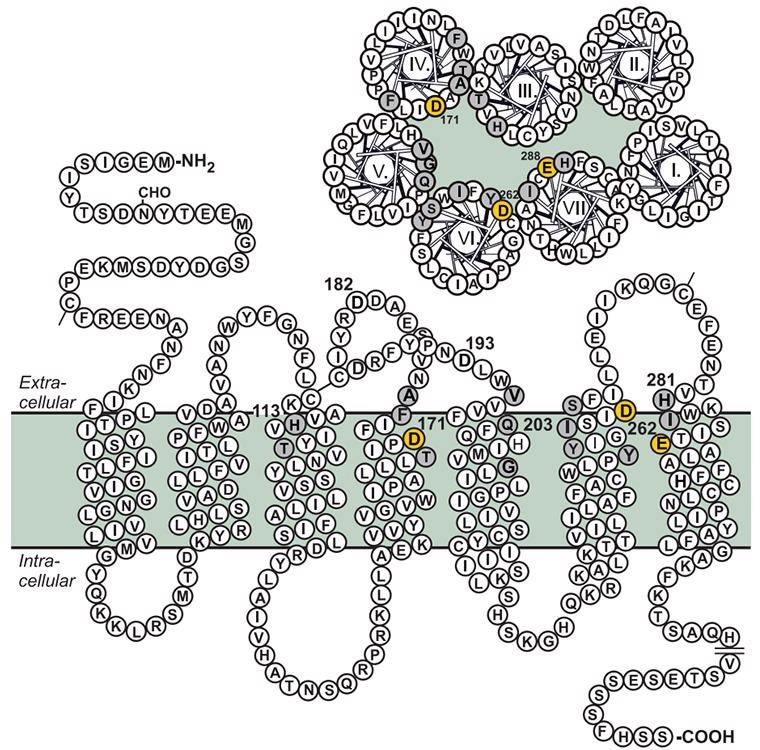
Structure of CXCR4. Image from reference
By attaching to the specific ligand CXCL12 (SDF), CXCR4 plays a key role in embryo development, cell migration, angiogenesis, immune response, stem cell homing, inflammatory response, tumor growth, and metastasis.
CXCR4 knockout mice exhibited embryonic lethality and defects in vascular development, hematopoiesis, and Cardiac defect, further highlighting the significant physiological role of CXCR4.
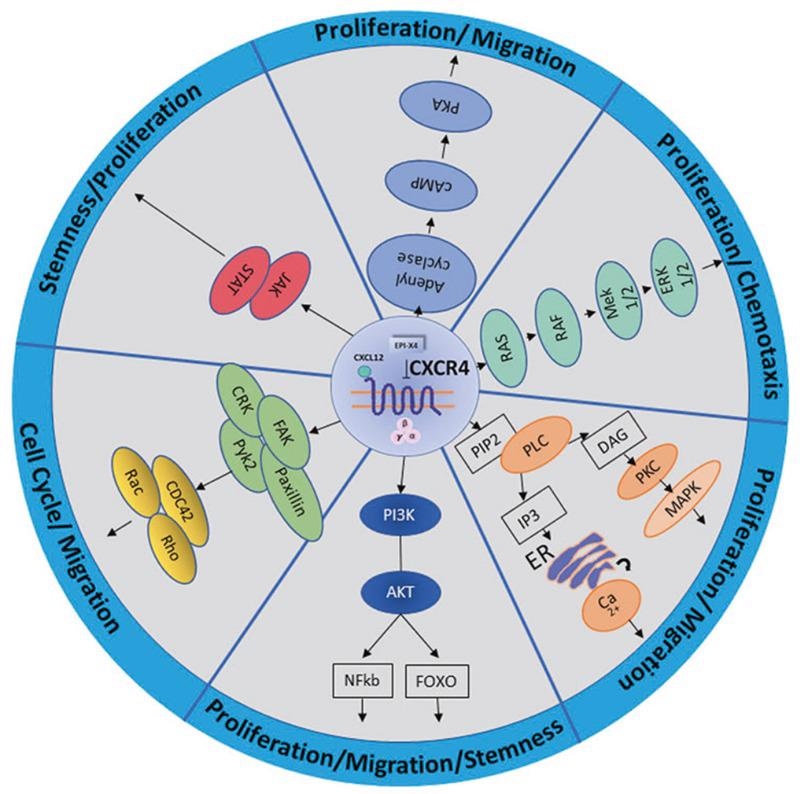
Signaling pathway of CXCR4. Image from reference
CXCR4 functions as an HIV co-receptor by the binding CD4 protein, allowing HIV to enter cells. As illustrated in the image below, gp120 on the HIV viral membrane is essential for infecting CD4 T cells. When gp120 unites to the CD4 receptor, it must also connect to a co-receptor like CXCR4 in order to enter the cell. As a result, CXCR4 antagonists are frequently used in HIV treatment.
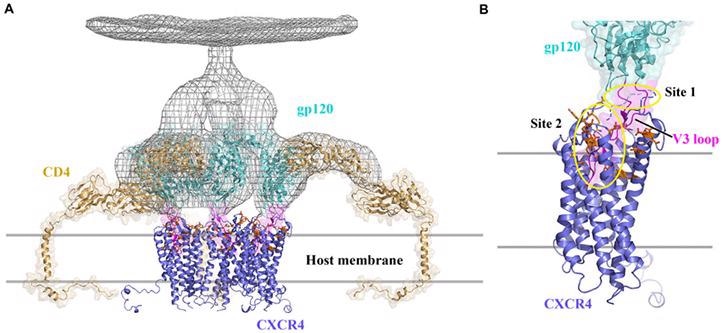
Structural model of CXCR4 interacting with CD4 to mediate HIV-1 entry into cells. Image from reference
Since CXCR4 can govern hematopoietic stem cell migration, it may play a significant role in clinical bone marrow hematopoietic stem cell transplantation. Furthermore, CXCR4 is strongly present in a variety of cancers, and its interpretation has been identified in 23 kinds of cancer.
CXCR4 overexpression in tumor cells promotes tumor growth, invasion, angiogenesis, metastasis, recurrence, and resistance. CXCR4 antagonists have been shown to destabilize tumor-stromal interactions, make cancer cells more susceptible to cytotoxic drugs, and slow tumor growth and metastatic burden. As a result, targeting CXCR4 is regarded as a promising treatment.
Several CXCR4-targeting drugs are currently in the works, including small molecular antagonists, antibodies, gene therapy, and CAR-T therapy. Plerixafor (Mozobil), a small molecule antagonist that targets CXCR4, is now commercially available. Bristol-Myers Squibb’s antibody-drug ulocuplumab is presently in Phase II clinical experiments. This medication is used to treat leukemia, multiple myeloma, and WHIM syndrome.
Introduction to CCR5
CCR5 (C-C motif chemokine receptor 5) is a member of the chemokine receptor and GPCR families and is primarily expressed by T cells and macrophages, like CXCR4. It raises intracellular calcium numbers by stimulating downstream G proteins and other signaling molecules, thus also transmitting signals when merged with chemokine ligands.
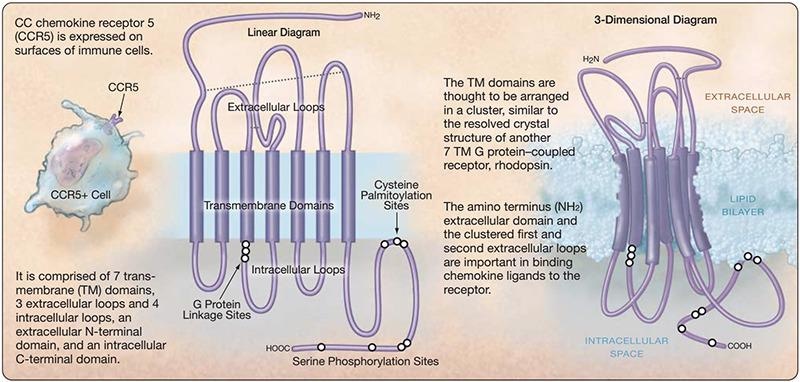
Structure of CCR5. Image from reference
CCR5 is a key co-receptor for HIV entry into host cells, and this gene defect is associated with HIV infection tolerance. CCR5 is thus a good target for HIV treatment. On February 15th, 2022, the “New York patient” became the third person in the world to achieve long-term AIDS remission.
A Weill Cornell Medical College research group showed that the incidence at the 29th Conference on Retroviruses and Opportunistic Infections (CROI) in Denver, USA: a New York City patient with HIV and acute myeloid leukemia (AML) has become the world’s first woman and third person to be cured of AIDS to date. The patient had garnered CCR5 32/32 stem cell transplantation from an HIV-resistant donor.
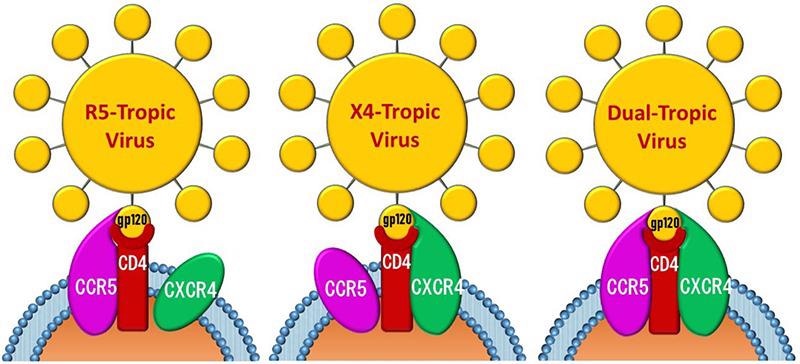
Chemokine receptors (CCR5 and CXCR4) mediate HIV entry into cells. Image from reference
Small molecule antagonists targeting CCR5 stop HIV from interacting with the receptor by binding to hydrophobic residues of CCR5. Maraviroc developed by Pfizer and GSK is the very first small-molecule anti-HIV oral drug, a CCR5 receptor antagonist, with the trade name Selzentry.
On August 6th, 2007, the FDA approved the drug, followed by the EMA on September 18th, 2007, and the PMDA on December 25th, 2008. Aside from CCR5 small molecule antagonists, monoclonal antibodies, gene therapy, and T cell medication for HIV treatment targeting CCR5 are all in the works.
Moreover, CCR5 is abundantly expressed in tumors and plays a major role in tumor initiation and progression. CCR5 is also a promising new therapeutic target for the treatment of advanced cancer.
ACROBiosystems is concentrated on meeting the obstacles associated with GPCR preparation. To support the customers and collaborators with antibody drugs and treatment interventions, designers provide full-length GPCR proteins through “FLAG” technology solutions. Full-length CCR5 and CXCR4 proteins have recently been developed and published.
Product list
Source: ACROBiosystems
| Molecule |
Cat. No. |
Species |
Product Description |
| CXCR4 |
CX4-H5219 |
Human |
Human CXCR4 / CD184 Full Length Protein-VLP (HEK293)NEW |
| CCR5 |
CC5-H52P3 |
Human |
Human CCR5 Full Length Protein-VLP (HEK293)NEW |
Product features
- Full-length CCR5 and CXCR4 proteins are with complete and native conformation
- Sophisticated immunogenicity due to inherent characteristics of VLP
- Excellent biological activity verified by binding to antibodies
- 100-300 nm in size and high identity confirmed by DLS, can be used as an ideal target for dendritic cells and phage display
- Suitable for immunization/ELISA/SPR/BLI/cell experiment etc.
Verification data
Full-length CXCR4-VLP Protein (Cat. No. CX4-H5219)
- High bioactivity verified by ELISA
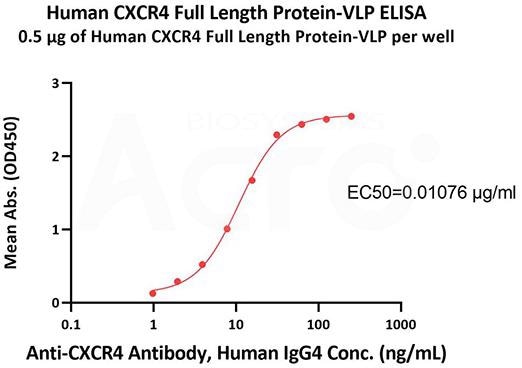
Immobilized Human CXCR4 Full-Length Protein-VLP (Cat. No. CX4-H5219) at 5 μg/mL (100 μL/well) can bind Human ulocuplumab, Human IgG4 with a linear range of 1-31 ng/mL (QC tested). Image Credit: ACROBiosystems
- High identity verified by DLS
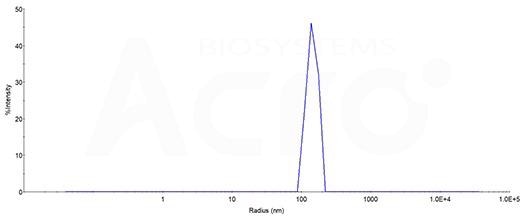
The mean peak Radius of CXCR4-VLP is 110-170 nm with more than 95% intensity as determined by dynamic light scattering (DLS). Image Credit: ACROBiosystems
Full-length CCR5-VLP Protein (Cat. No. CC5-H52P3)
- High bioactivity verified by ELISA
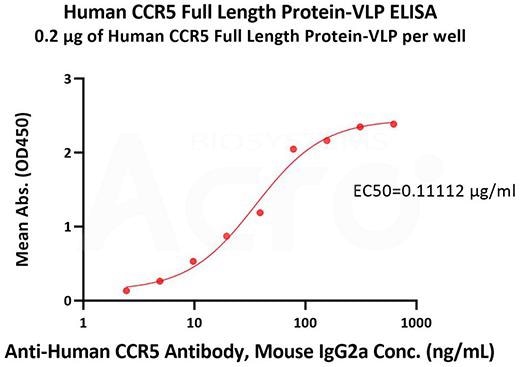
Immobilized Human CCR5 Full-Length Protein-VLP (Cat. No. CC5-H52P3) at 2 μg/mL (100 μL/well) can bind Anti-Human CCR5 Antibody, Mouse IgG2a with a linear range of 2-78 ng/mL (QC tested). Image Credit: ACROBiosystems
- High identity verified by DLS
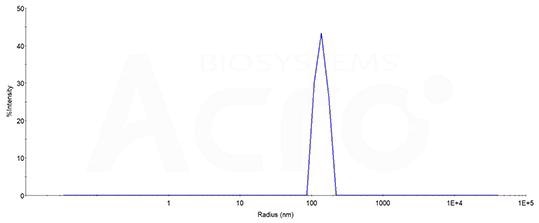
The mean peak Radius of CCR5-VLP is 130-170 nm with more than 95% intensity as determined by dynamic light scattering (DLS). Image Credit: ACROBiosystems
References
- Guido Lancman1, Dahniel L. Sastow, et al. Bispecifc Antibodies in Multiple Myeloma: Present and Future. Blood Cancer Discov 2021, 2:423–33. doi: 10.1158/2643-3230.BCD-21-0028
- Lederman MM, Penn-Nicholson A, Cho M, Mosier D. Biology of CCR5 and its role in HIV infection and treatment. JAMA. 2006, 296(7):815-26. doi: 10.1001/jama.296.7.815. PMID: 16905787.
- Fedora Grande, Maria Antonietta Occhiuzzi, Bruno Rizzuti et al. CCR5/CXCR4 Dual Antagonism for the Improvement of HIV Infection Therapy. Molecules. 2019, 24(3), 550; https://doi.org/10.3390/molecules24030550
- Blanpain C, Migeotte I, Lee B, Vakili J, Doranz BJ, Govaerts C, Vassart G, Doms RW, Parmentier M. CCR5 binds multiple CC-chemokines: MCP-3 acts as a natural antagonist. Blood. 1999, 94(6):1899-905. PMID: 10477718.
- Tagliamonte M, Tornesello ML, Buonaguro FM, Buonaguro L. Conformational HIV-1 envelope on particulate structures: a tool for chemokine coreceptor binding studies. J Transl Med. 2011, 27; 9 Suppl 1(Suppl 1): S1. doi: 10.1186/1479-5876-9-S1-S1. PMID: 21284899; PMCID: PMC3105500.
- Barmania F, Pepper MS. C-C chemokine receptor type five (CCR5): An emerging target for the control of HIV infection. Appl Transl Genom. 2013(2):3-16. doi: 10.1016/j.atg.2013.05.004. PMID: 27942440; PMCID: PMC5133339.
- Jiao X, Nawab O, Patel T, Kossenkov AV, Halama N, Jaeger D, Pestell RG. Recent Advances Targeting CCR5 for Cancer and Its Role in Immuno-Oncology. Cancer Res. 2019, 79(19):4801-4807. doi: 10.1158/0008-5472.CAN-19-1167. Epub 2019 Jul 10. PMID: 31292161; PMCID: PMC6810651.
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.