Featuring a similar structure to the MHC class I molecule, the neonatal Fc receptor is an Fc receptor that forms a heterodimer protein with beta-2-microglobulin. It was initially identified in rodents as a receptor that can transport IgG across a newborn’s gut into the bloodstream. For this reason, it was later named a 'neonatal' Fc receptor (FcRn). Additional studies have also detected analogous receptors in humans and other mammals.
The primary function of the FcRn is to prolong the half-life of serum albumin and IgG by lowering lysosomal degradation in endothelial cells. As indicated in Figure 1, FcRn binds serum albumin and IgG under acidic pH (<6.5) conditions, allowing them to be recycled to the surface of the cell, where they are released into the blood at a neutral pH (>7.0) level. This mechanism serum albumin and IgG from lysosomal degradation.
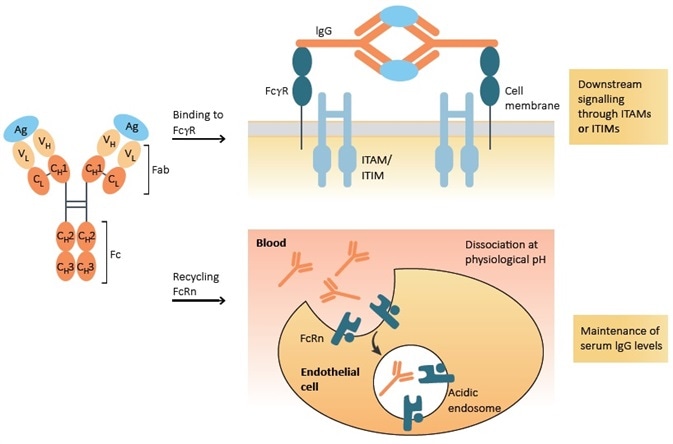
Figure 1. Function of Fc receptor
Besides FcRn, there are three more classes of Fc gamma receptors (FcγRs) that can interact with human IgG. These types of receptors are differentially expressed by macrophages, monocytes, eosinophils, neutrophils, B and T cells, natural killer cells, platelets, and dendritic cells. Fc γ Rs in humans bind IgG with different affinities that range from low (>10-7 M, Fc γ RII, or CD32), medium (<10-7 M, Fc γ RIII, or CD16), to high (10-8‒10-9 M, Fc γ RI, or CD64).
Furthermore, varied IgG isotypes exhibit different binding affinities to these receptors. Interaction occurs between IgG and Fc-gamma receptors, mediating a wide range of cellular function, especially antibody-dependent cell-mediated cytotoxicity (ADCC).
Polymorphisms of the Fc γ Rs are quite common and can impact mAb binding. Certain Fc γ R polymorphism has been associated with clinical outcomes. For instance, a clinical examination of rituximab (anti-CD20) in the treatment of non-Hodgkin’s lymphoma has demonstrated a higher response rate among patients who have Fc γ RIIIa (V176) homozygous, as opposed to those with F176 homozygous and F176/V176 heterozygous.
An analysis of the side effects of OKT3—the mouse monoclonal anti-CD3 antibody—is another example of polymorphism. The anti-CD3 antibody is commonly used as an immunosuppressive agent to prevent the rejection of solid-organ transplants. It was shown that the severity of this “first-dose reaction” was associated with the presence of the Fc γ RIIa (R167) allotype, which interacts with mIgG1.
The interaction of therapeutic antibodies with FcRn and Fc-gamma receptors decides their efficacy. Thus, candidates must be tested against a series of receptors at the time of antibody engineering. ACROBiosystems provides a detailed collection of recombinant Fc receptor proteins, which also include their common variants, to speed up research times.
Key Features of Product
High Purity
In order to fulfill the high purity needs of pharmaceutical applications, ACROBiosystems’ production team carries out SDS-PAGE as well as HPLC analyses of the Fc receptors for quality control (QC) purposes. It should be noted that the monomeric Fc receptor should be used, which represents its natural state.
The formation of oligomers at the time of production may lead to artificially improved binding interaction as a result of the avidity effect. In order to prevent such situations, rigorous internal control standards of monomer purity have been established by HPLC testing. Only batches that satisfy all purity requirements are released.
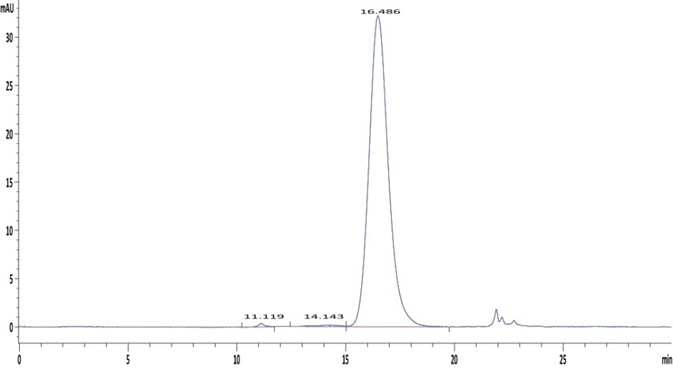
Figure 2. The purity of Human Fc gamma RIIA/CD32a (R167) Protein (Cat. No. CDA-H5221) is greater than 95% as determined in a HPLC analysis. Image Credit: ACROBiosystems
Bioactivity Validated by SPR
Many methods can be used to determine the binding affinity between two molecules. Yet, the studies of Fc interaction are usually performed by SPR, because conventional ELISA is unsuitable for low-affinity receptors (for example, CD32 and CD16).
To ensure that ACROBiosystems’ Fc receptors deliver optimal results, the company’s QC team employs Biacore platform to test the products against reference antibodies. All protocols for SPR are provided for free.
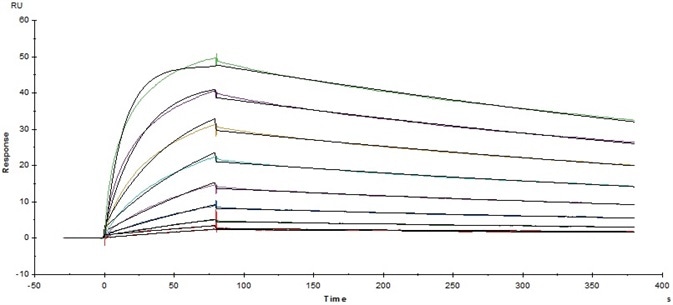
Figure 3. Immobilized Human Fc gamma RI/CD64, His Tag (Cat. No. FCA-H52H2) on CM5 Chip via anti-His antibody, can bind Herceptin with an affinity constant of 5.45 nM as determined in a SPR assay (Baicore T200). Image Credit: ACROBiosystems
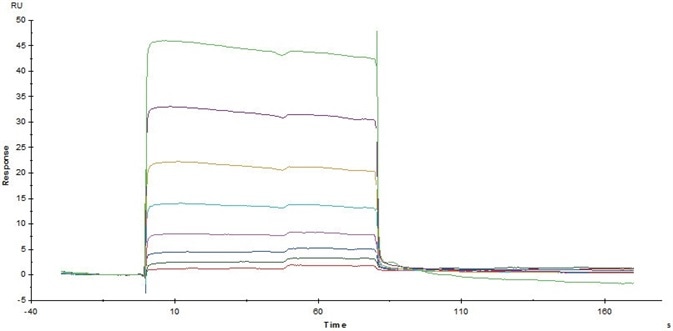
Figure 4. Immobilized Human CD32a (R167) (Cat. No. CDA-H5221) on CM5Chip via anti-His antibody, can bind Rituximab with an affinity constant of 3.12 μM as determined in SPR assay (Biacore T200). Image Credit: ACROBiosystems
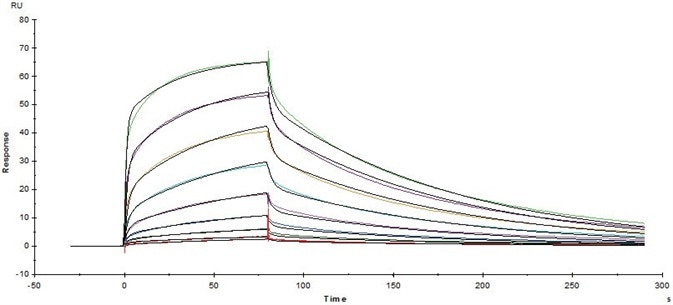
Figure 5. Immobilized Human CD16a (V176) Protein (Cat. No. CD8-H52H4) on CM5Chip via anti-His antibody, can bind Herceptin with an affinity constant of 163 nM as determined in SPR assay (Biacore T200). Image Credit: ACROBiosystems
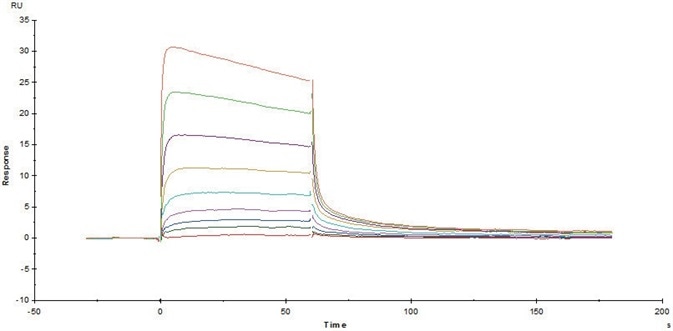
Figure 6. Immobilized Human FcRn/FCGRT & B2M Heterodimer Protein (Cat. No. FCM-H5286) on CM5 Chip via anti-His antibody, can bind Herceptin with an affinity constant of 0.489 μM as determined in a SPR assay (Biacore T200). Image Credit: ACROBiosystems
High Batch-to-Batch Consistency
ACROBiosystems follows stringent quality control measures to guarantee the best performance of its product. As indicated below, the batch difference among the tested samples is insignificant.
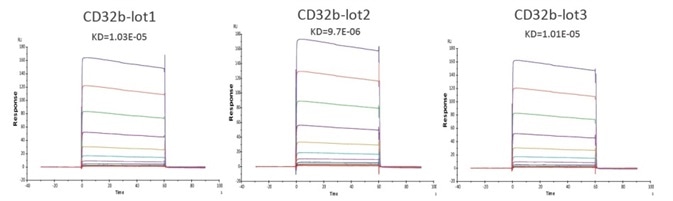
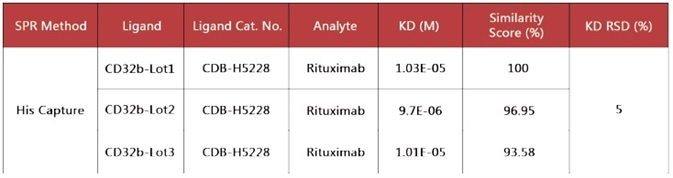
Figure 7. Immobilized Human Fc gamma RIIB/CD32b Protein (Cat. No. CDB-H5228) on CM5 Chip via anti-His antibody, can bind Rituximab with an affinity constant of 10 μM as determined in a SPR assay (Biacore T200). Image Credit: ACROBiosystems
The performance of different batches of CD32b (CDB-H5228) in the same assay have been compared, and the software analysis demonstrated that the similarity score was very high, indicating high batch-to-batch consistency.
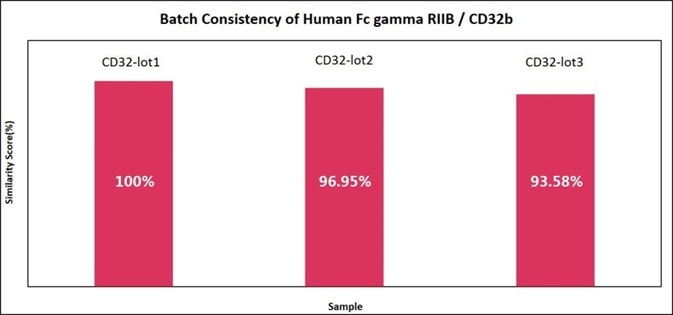
Figure 8. Batch consistency of Human Fc gamma RIIB/CD32b (Cat. No. CDB-H5228). The Similarity for different batches is more than 90%. Image Credit: ACROBiosystems
Biotinylated or Other Species Fc Receptors Available
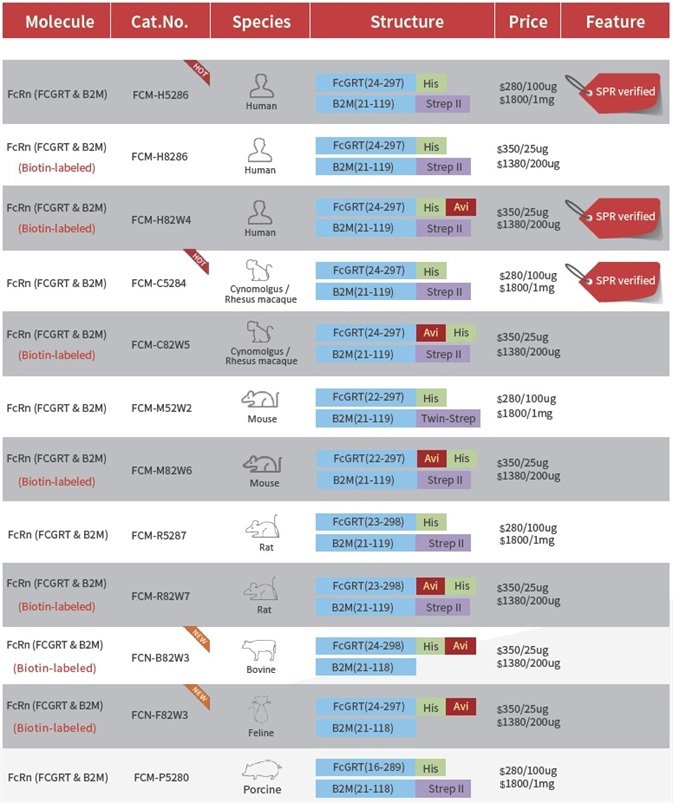
When biotin labeling is used, it can make the assay development relatively easier. ACROBiosystems provides a range of ready-to-use biotinylated Fc receptors that are made using its in-house developed labeling methods, which confer minimal batch-to-batch variation and high bioactivity. The company has also produced a series of Fc receptors of other species, which can be used for the screening of the species cross-reaction or non-humanized antibodies.
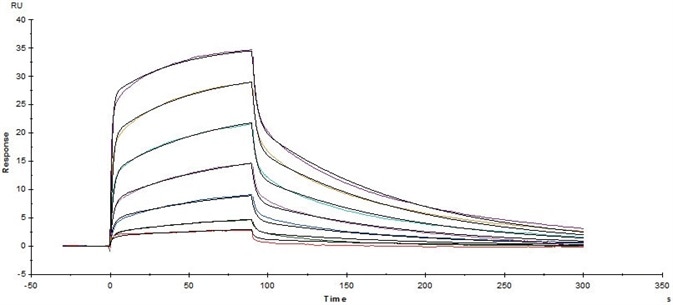
Figure 9. Immobilized Biotinylated Human CD16a (V176), His Tag, Avi Tag(Cat. No. CDA-H82E9) on Series S Sensor Chip CAP via Biotin Capture can bind Rituximab with an affinity constant of 0.261 μM as determined in a SPR assay (Biacore T200). Image Credit: ACROBiosystems
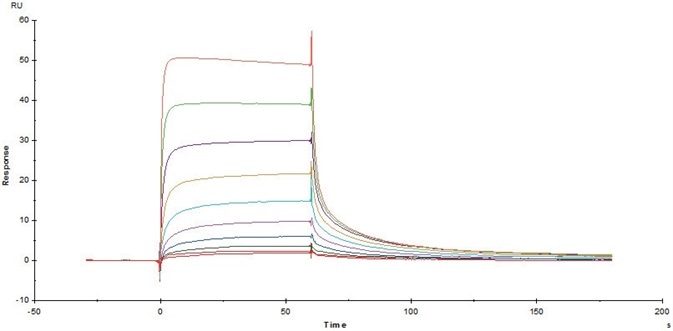
Figure 10. Immobilized Cynomolgus/Rhesus macaque FcRn Protein (Cat. No. FCM-C5284) on CM5 Chip via anti-His antibody, can bind Herceptin with an affinity constant of 0.403 μM as determined in a SPR assay (Biacore T200). Image Credit: ACROBiosystems
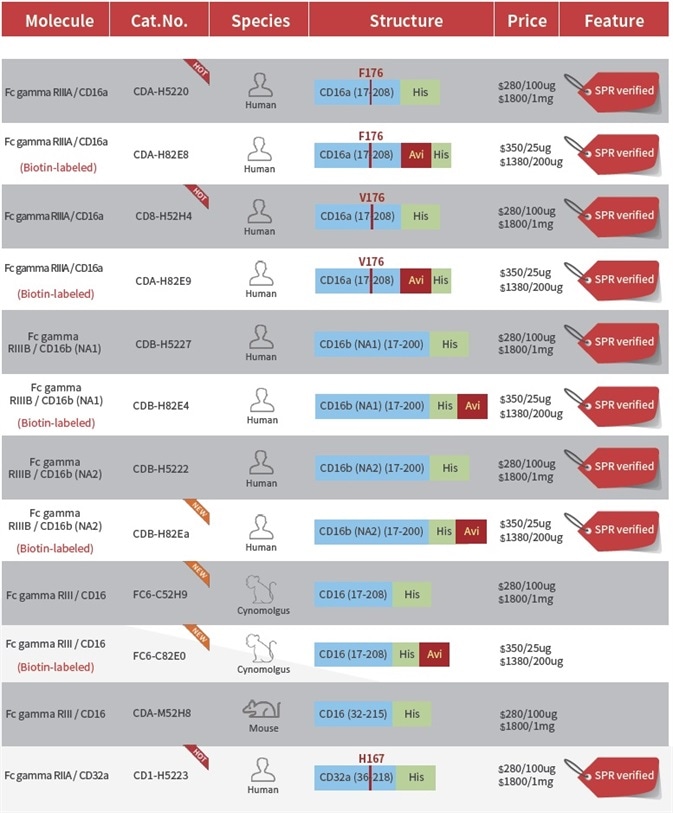
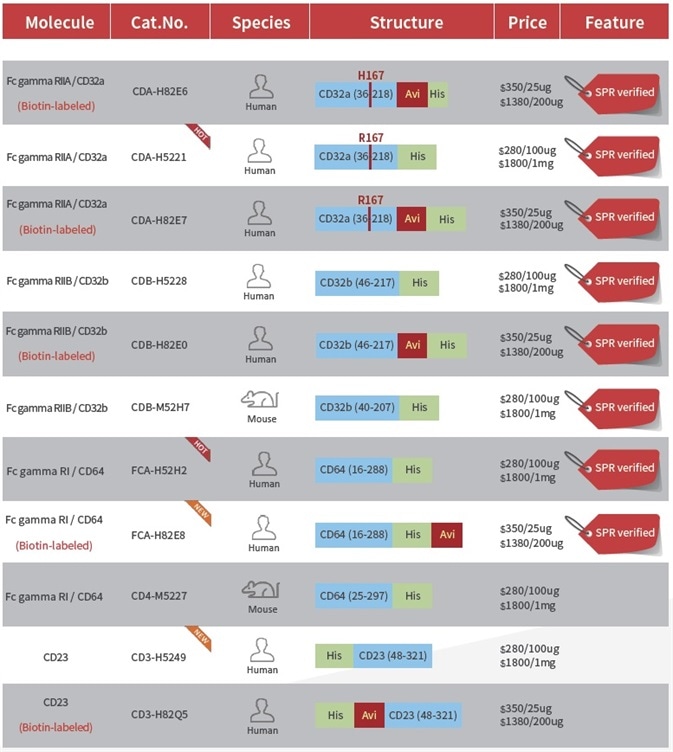
Product List
ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.