CAR-T cell therapy offers the hope of a cure to a number of cancer patients. Yet, in spite of the great effectiveness, more attention has been paid to the toxicity of the therapy known as cytokine release syndrome (CRS).
This article is aimed at summarizing the main points of assessment, grading and management of CRS in CAR-T Therapy and provide answers to the following questions:
- How to identify the onset of CRS clinically?
- What is CRS management?
- What is the ASTCT CRS consensus grading?
- How to use tocilizumab in CRS management?
Assessment, grading and management of CRS in CAR-T therapy
In recent times, many cancer patients are hopeful for a cure in relation to CAR-T cell therapy. However, even though the treatment is extremely effective, the inherent toxicity of cytokine release syndrome (CRS) is a significant obstacle.
Subsequent to the administration of CAR-T into the human body, T lymphocytes are activated and rapidly proliferate, which leads to an excessive release of cytokines including TNF-α, IFN-γ, IL-1, IL-2, IL-4, IL-6, IL-8, IL-10 and IL-12.
This effect can lead to complications including fever, hypotension, blood coagulation disorders, dyspnea, myalgia and end-organ dysfunction. The result could mean permanent damage to tissues and organs and be fatal in severe cases.
To summarize, CRS is an acute systemic inflammatory syndrome that is associated with CAR-T cell therapy [1]. However, there are many influencing components that can influence the occurrence of CRS, including treatment procedure, disease state and drugs [2]. (Table 1)
Table 1. Risk factors for CRS. Source: ACROBiosystems

How to identify the onset of CRS clinically?
CRS patients usually have levels of serum CRP and ferritin that are higher than normal.
They can be used as biomarkers to identify the development of CRS. Yet, these markers, including ferritin, CRP, LDH, AST, ALT and creatinine only appear after the clinical manifestations of CRS. Thus, it is not possible to determine the onset of CRS using these biomarkers.
A majority of hospitals cannot monitor real-time cytokine levels; they can only detect the clinical manifestations. Factors that include the type of CAR-T therapy and the disease state and condition of the patient typically decide when the CRS is going to occur.
For patients taking Kymriah, which contains the costimulatory domain of 4-1BB (CD137), the peak of CRS happens on the 3rd and 7th day after administration.
However, for patients taking Yescarta or Tecartus, which include the costimulatory domain of CD28, the CRS can occur earlier, usually on the second day of administration. But a number of delayed CRS cases were reported three weeks after administering CAR-T administration.[2]
In 2019, the American Society for Transplantation and Cell Therapy (ASTCT) drew up a grading guideline for CRS consensus (Table 2). The guideline stipulates that the presence of a fever (≥38°C) is a requirement: with or without hypotension and/or hypoxia, the conditions can become more severe.
Table 2. ASTCT CRS Consensus Grading. Source: ACROBiosystems
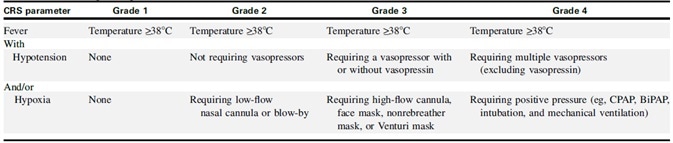
Studies have demonstrated that the ASTCT grading system can offer a rapid, accurate, objective and repeatable clinical assessment of the severity of CRS.[2-3]
Grade 1) CRS patients showing fever and other non-specific symptoms but no hypotension or hypoxia are placed into a grade 1 category. The strategy for treatment is intravenous infusion of antipyretic drugs.
Grade 2) Defined as CRS grade 2 patients show fever (≥38.0°C) with hypotension, yet not requiring vasopressors and/or hypoxia necessitating the use of oxygen supplied via low-flow nasal cannula (≤6 L/minute) or blow-by.
However, it crucial to note that CRS patients are at high risk of edema as a result of vasodilatation and capillary leakage. Thus, fluid infusion must be carefully monitored. Tocilizumab, which inhibits IL-6, can be issued to grade 2 patients.
Studies have demonstrated that early administration of tocilizumab can reduce the severity of CRS and organ failure while conserving the efficacy of CAR-T therapy.
Grade 3) For patients showing signs of grade 3 CRS, that is, patients that have fever, low blood pressure and no response to fluid infusion, while requiring high-flow oxygen (≥6L/min), admission to the intensive care unit (ICU) should be considered.
Condition management and careful use of Belizumab and glucocorticoids (dexamethasone, etc.) for treatment, and it is recommended to use vasopressor (such as norepinephrine) and/ or high-flow nasal cannula (>6 L/minute) in order to prevent visceral damage and further deterioration.
Additionally, as high-grade CRS patients are susceptible to cardiac dysfunction, it is advised to continuously monitor cardiac function.
Grade 4) This grade of CRS is defined as fever (≥38.0°C) with hypotension which necessitates several vasopressors (excluding vasopressin) and/or hypoxia requiring positive pressure (e.g., CPAP, intubation, bilevel positive airway pressure, mechanical ventilation) not ascribed to another cause.
It is recommended to use methylprednisolone at a dose of 1g/day, in addition to following the steps of the grade 3 CRS treatment. Studies show that with early intervention, high-grade CRS is reversible.
In 2017, the FDA approved the drug Roche's tocilizumab (trade name Actemra): developed for the CRS caused by CAR-T therapy through the blockage of the inflammatory cytokine interleukin-6 (IL -6).
Clinical guidance of tocilizumab [4]:
(1) Tocilizumab should be considered in the early stage of CRS for patients with severe complications or the elderly.
(2) Tocilizumab can be administered for children with grade 3 CRS and adults with grade 2 CRS.
(3) Tocilizumab can be administered to pediatric patients who show grade 2 CRS but are intolerant to fever.
(4) If the conditions do not improve for adults and children administered with one dose of tocilizumab, the second dose should be combined with glucocorticoids. If there is still no improvement in the patient’s condition, consider Anakinra, Siltuximab and a high dose of methylprednisolone.
Researchers are grafting hard to find preventative solutions or appropriate treatment for CRS. In 2018, Nature published two articles on the same day that discussed a new way to control CAR-T toxicity.
The research demonstrated that the inhibition of IL-1 limits the macrophage-induced CRS. In 2019, Michel Sadelain, director of the Cell Engineering Center of Sloan Kettering Cancer Center, published an article printed in Science translational medicine.
Sadelain determined that a functional switch was present in CAR-T cells, which prevented incidents of CRS in mouse models.[5] In January 2020, an article in Science Immunology was put forward by a research team from the Chinese Academy of Medical Sciences.
This article presented the process of pyrolysis as a result of CAR-T therapies and CRS’s mechanisms. From the clarity of the microscope images, it is possible to observe the entire process of CRS triggered by CAR-T cell therapy.[6]
With the ongoing clinical research of CAR-T therapies and CRS, breakthroughs are expected in the near future.
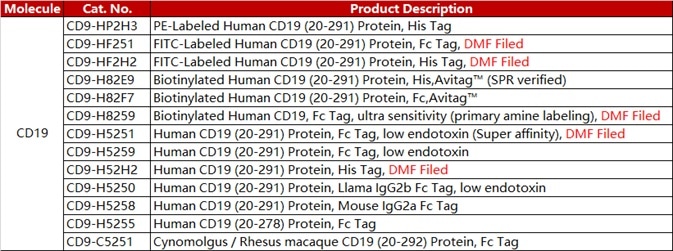
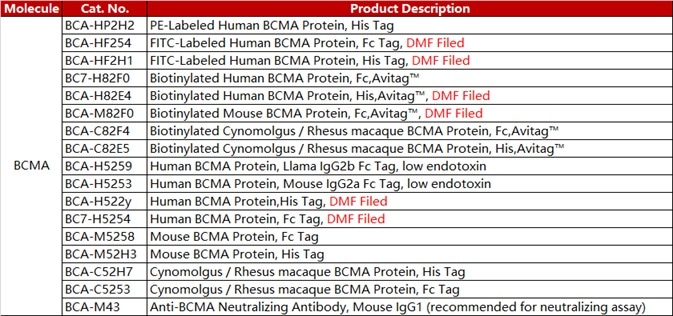
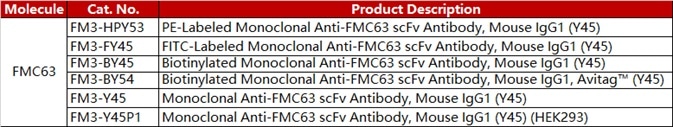
ACROBiosystems developed a series of CAR-T target proteins, including CD19, BCMA, CD22 MSLN and EGFR. We have all these proteins in different forms like non-labeled, biotin-labeled and fluorescent-labeled, etc. We also provide the flow cytometry protocols for CAR expression detection.
ACRO FITC-labeled CD19 protein (Cat. No. CD9-HF2H2) is exceptionally specific and sensitive in anti-CD19 CAR detection assays. Another antibody (Cat. No. FM3-FY45) was developed to target the antigen-binding epitope of FMC63 scFv. This product facilitates an additional technique for Anti-CD19 (FMC63) CAR detection.
ACRO FITC-labeled BCMA protein (Cat. No. BCA-HF254) offers excellent sensitivity. This product has been used extensively across a variety of assays and is well-known throughout the industry.
Product list
CD19
BCMA
Anti-FMC63 mAb
Case study
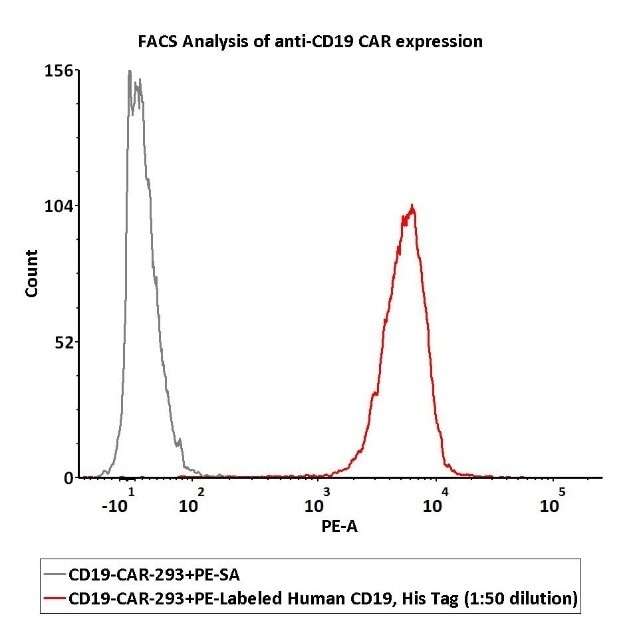
Image Credit: ACROBiosystems
Staining 1e6 of the anti-CD19 CAR-293 cells with 100 μL of 1:50 dilution (2 μL stock solution in 100 μL FACS buffer) of PE-Labeled Human CD19 (20-291), His Tag (Cat. No. CD9-HP2H3). PE Streptavidin was utilized as a negative control.
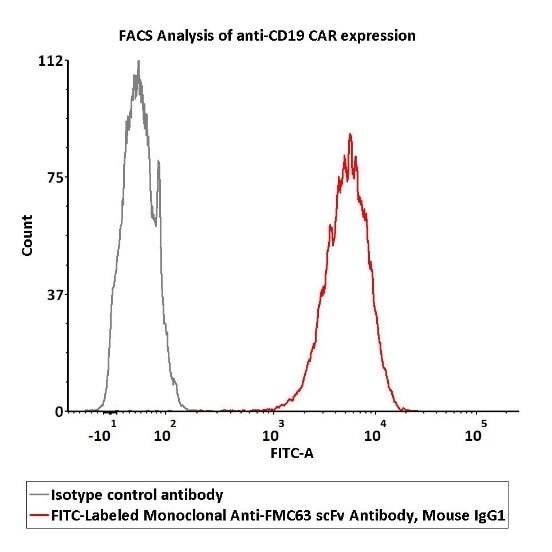
Image Credit: ACROBiosystems
2e5 of Anti-CD19 CAR-293 cells were stained with 100 μL of 1:50 dilution (2 μL stock solution in 100 μL FACS buffer) FITC-Labeled Monoclonal Anti-FMC63 scFv Antibody, Mouse IgG1 (Cat. No. FM3-FY45) and isotype control sequentially. FITC signal was utilized to assess the binding activity.
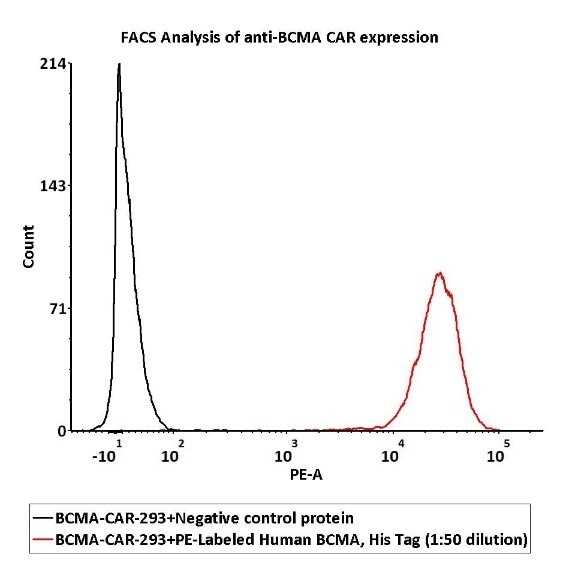
Image Credit: ACROBiosystems
PE signal was utilized to evaluate the binding activity while 1e6 of the Anti-BCMA CAR-293 cells were stained with 100 μL of 1:50 dilution (2 μL stock solution in 100 μL FACS buffer) of PE-Labeled Human BCMA Protein, His Tag (Cat. No. BCA-HP2H2) and negative control protein respectively.
References
- https://www.cn-healthcare.com/articlewm/20190306/content-1047067.html .
- Schubert ML, Schmitt M, Wang L, et al. Side-effect management of chimeric antigen receptor (CAR) T-cell therapy. Ann Oncol. 2021;32(1):34-48.
- Freyer CW, Porter DL. Cytokine release syndrome and neurotoxicity following CAR T-cell therapy for hematologic malignancies. J Allergy Clin Immunol. 2020;146(5):940-948.
- Maus MV, Alexander S, Bishop MR, et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune effector cell-related adverse events. J Immunother Cancer. 2020;8(2):e001511.
- Mestermann, Katrin, et al. "The tyrosine kinase inhibitor dasatinib acts as a pharmacologic on/off switch for CAR T cells." Science translational medicine 11.499 (2019).
- Liu, Yuying, et al. "Gasdermin E–mediated target cell pyroptosis by CAR T cells triggers cytokine release syndrome." Science immunology 5.43 (2020).
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.