The interleukin-4 receptor (IL-4R) has become an “inspiring” target for cutting-edge treatments for atopic diseases. This type-1 transmembrane protein is essential to the molecular mechanism that underlies the type-2 inflammations, a particular pattern of the immune response.

Image Credit: ACROBiosystems
Atopic dermatitis (AD) and specific forms of asthma are two medical disorders that are mostly brought on by these inflammations. To fully comprehend the mechanism of therapeutic action and appreciate the enormous potential of IL-4R alpha targeting medicines, it is essential to have a basic understanding of the molecular process behind this immune response pattern.
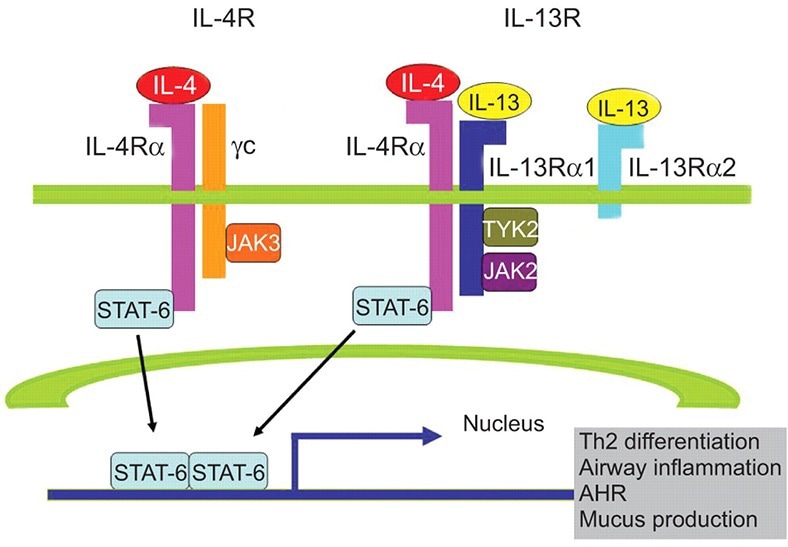
IL-4R cellular pathway. Image Credit: ACROBiosystems
The biological and immunological functions of B-lymphocytes, monocytes, dendritic cells, and fibroblasts are all affected by the cytokines IL-4 and IL-13. These cytokines interact with the IL-4 and IL-13 receptors to start the type 2 inflammatory pathway, which results in Th2 cell differentiation, airway inflammation, and mucus production.
The type-2 inflammatory pathway is first activated in allergic illnesses like AD by aberrant cytokine release resulting from an imbalance of Th1 and Th2 differentiation. Th2 cells that have been activated release cytokines, including IL-4, 13, and 31, which prompt downstream B cells to undergo a change and produce IgE antibodies.
Mast cells and basophils are then called upon to degranulate and release inflammatory substances. Simultaneously, the secreted IL-4 and 13 continue to bind to its respective receptors, such as IL-4R, that repeatedly promotes TH2 differentiation and subsequent inflammation.
The type-2 inflammation pathway’s signaling role for IL-4 and IL-13 identifies IL-4R alpha as a potential target for individuals who exhibit aberrant type-2 inflammatory responses. Contrary to medications that target IL-4 and IL-13 exclusively, IL-4R alpha has a novel mechanism resembling “bi-specific” targets.
The immune system is controlled by antibodies that block the binding of both IL-4 and IL-13, which inhibits intracellular signaling and reduces the production of IgE, Th2 cell differentiation, and inflammatory substances. As a result, IL-4R targeting medication research has received a lot of attention from pharmaceutical companies all over the world.
Table 1. Advances in drug research and development targeting IL-4R alpha. Source: Pharmacodia
| Drug Name |
Highest Stage |
Company |
Indications |
| Dupilumab |
Launched (2017) |
Sanofi/Regeneron |
Eosinophilic Esophagitis; Atopic Dermatitis;
Sinusitis; Nasal Polyps; Asthma |
| CM-310 |
Phase III |
Keymed Biosciences
CSPC Pharmaceutical |
Sinusitis; Nasal Polyps; Atopic Dermatitis; Pruritus;
Solid Tumors; Asthma; Nasal Disorders; Allergic Rhinitis |
| Recombinant anti-IL-4Rα humanized monoclonal antibody |
Phase II |
Sunshine Guojian |
Atopic Dermatitis |
| MG-K10 |
Phase II |
MABGEEK
Dragon Boat Pharmaceutical |
Atopic Dermatitis; Asthma |
| Manfidokimab |
Phase II |
Akeso Pharmaceuticals |
Atopic Dermatitis; Asthma |
| QX-005N |
Phase II |
Qyuns Therapeutics |
Atopic Dermatitis; Prurigo; Asthma;
Chronic Urticaria; Sinusitis |
| Elarekibep |
Phase II |
AstraZeneca
Pieris Australia Pty Ltd |
Asthma |
| CBP-201 |
Phase II |
Connect Biopharma |
Atopic Dermatitis; Asthma |
| SHR-1819 |
Phase I |
Atridia Pty Ltd;
Jiangsu Hengrui Pharmaceuticals |
Atopic Dermatitis; Asthma |
| LQ-036 |
Phase I |
Shanghai Novamab Biopharmaceuticals;
Syneos Health |
Asthma |
| BA2101 |
Preclinical |
Boan Biotech |
Atopic Dermatitis; Asthma;
Sinusitis;Itching; Hives |
The only IL-4R alpha targeting medication on the market right now is Dupilumab, which was created in collaboration between Sanofi and Regeneron. Dupilumab sales surpassed $4 billion in 2020, and by 2024, they are expected to reach over $10 billion. One of the main targets for Dupilumab is AD, otherwise known as eczema, which is a prevalent disease that affects millions of people.
According to epidemiologic data from Frost & Sullivan, there are currently 649 million AD patients worldwide, and that number is expected to rise to 755 million by 2050. Topical creams and ointments are currently used as the first-line treatment for moderate-to-severe AD in order to control the level of itching. However, Dupilumab prevents inflammation from emerging in patients with uncontrolled AD by blocking the inflammation signaling system.
Its effectiveness has resulted in a multi-disciplinary consensus statement that emphasizes the use of Dupilumab as a first-line systemic treatment modality when topical therapy does not prove effective. The success of Dupilumab in controlling severe cases of AD has helped broaden its clinical applications to other medical conditions, including sinusitis, eosinophilic esophagitis, and specific cases of asthma.
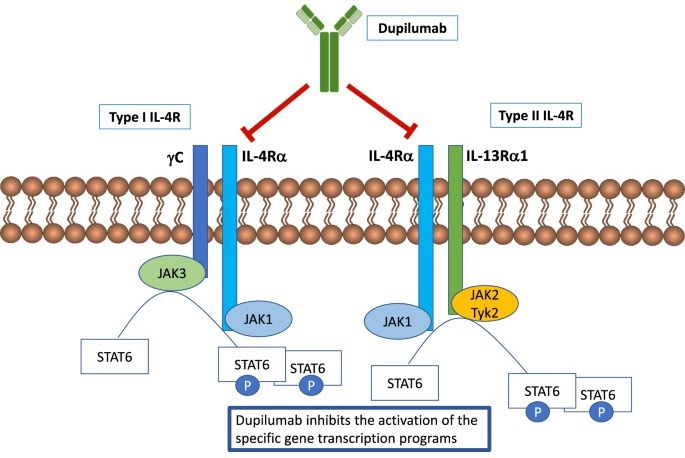
Mechanism of Action of Dupilumab. Image Credit: ACROBiosystems
Ultimately, the therapeutic target of IL-4R alpha is very promising for the control and treatment of atopic diseases. To aid in the development of IL-4R alpha targeted therapeutics, ACROBiosystems has introduced a variety of IL-4R alpha/IL-4/IL-13 recombinant proteins that are ideal for antibody screening and functional verification.
Product features
- Expressed by HEK293 cells — Recombinant proteins close to the native conformation
- Availability of different species — Human, Mouse, Cynomolgus/Rhesus macaque
- Numerous tags provided — His Tag, His & Avi Tag, Fc Tag, Fc & Avi Tag
- High bioactivity — Verified by ELISA binding and competition experiments
- High purity and structural homogeneity — Verified by SDS-PAGE and SEC-MALS
Product list
Table 2. Source: ACROBiosystems
| Molecule |
Cat. No. |
Species |
Product Description |
| IL-4 R alpha |
ILR-H5221 |
Human |
Human IL-4 R alpha / CD124 Protein, His Tag |
| ILR-H82E9 |
Human |
Biotinylated Human IL-4 R alpha / CD124 Protein, Avitag™, His Tag |
| ILR-H5253 |
Human |
Human IL-4 R alpha / CD124 Protein, Fc Tag |
| ILR-H82F4 |
Human |
Biotinylated Human IL-4 R alpha / CD124 Protein, Fc, Avitag™ |
| ILR-M5252 |
Mouse |
Mouse IL-4 R alpha / CD124 Protein, Fc Tag |
| ILR-M52H1 |
Mouse |
Mouse IL-4 R alpha / CD124 Protein, His Tag |
| ILR-C5258 |
Cynomolgus / Rhesus macaque |
Cynomolgus / Rhesus macaque IL-4 R alpha / CD124 Protein, Fc Tag |
| ILR-C52H8 |
Cynomolgus / Rhesus macaque |
Cynomolgus / Rhesus macaque IL-4 R alpha / CD124 Protein, His Tag |
| IL-4 |
IL4-H4218 |
Human |
ActiveMax® Human IL-4 Protein, Tag Free |
| IL4-H82E0 |
Human |
Biotinylated Human IL-4 Protein, Avitag™, His Tag |
| IL4-H52H9 |
Human |
Human IL-4 Protein, His Tag (MALS verified) |
| IL4-M52H5 |
Mouse |
Mouse IL-4 Protein, His Tag |
| IL4-C5259 |
Cynomolgus |
Cynomolgus IL-4 Protein, Fc Tag (MALS verified) |
| IL-13 |
IL3-H52H4 |
Human |
Human IL-13 Protein, His Tag |
| IL3-H5256 |
Human |
Human IL-13 Protein, Fc Tag |
| IL3-H82E5 |
Human |
Biotinylated Human IL-13 Protein, His, Avitag™ |
| IL3-C52H4 |
Canine |
Canine IL-13 Protein, His Tag |
IL-2 and IL-2 receptors
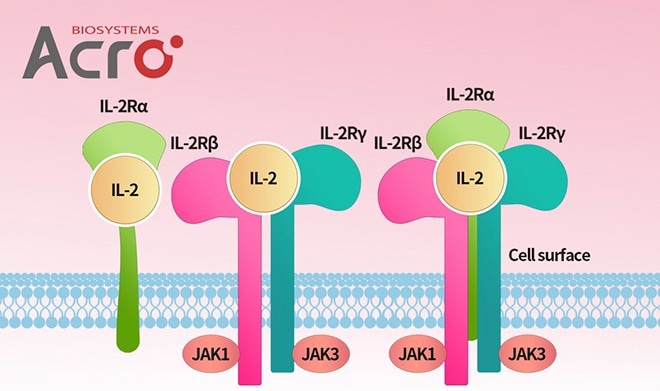
Image Credit: ACROBiosystems
- IL-2, IL-2 R βγ, IL-2R α, IL-2R αβγ
- Affinity verified by SPR (Biacore)
IL-6 and IL-6 receptors
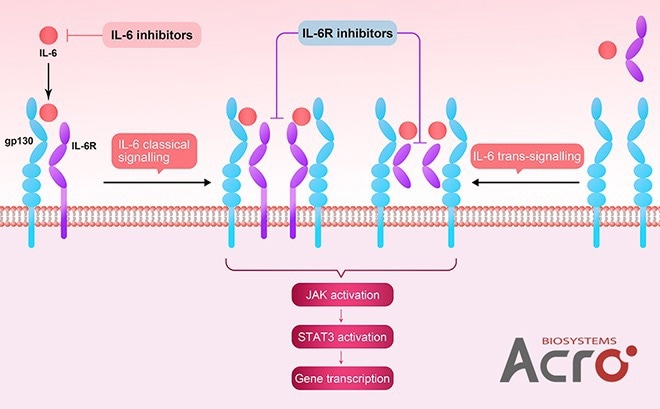
Image Credit: ACROBiosystems
- IL-6, IL-6 R alpha, gp130
- ELISA/BLI/cell-based assay verified

Image Credit: ACROBiosystems
References
- C. K. Oh, G. P. Geba, N. Molfino. Investigational therapeutics targeting the IL-4/IL-13/STAT-6 pathway for the treatment of asthma. European Respiratory Review 19, 46-54 (2010). DOI: 10.1183/09059180.00007609
- Licari, A., Castagnoli, R., Marseglia, A. et al. Dupilumab to Treat Type 2 Inflammatory Diseases in Children and Adolescents. Pediatr Drugs 22, 295–310 (2020). https://doi.org/10.1007/s40272-020-00387-2.
About ACROBiosystems
ACROBiosystems is a cornerstone enterprise of the pharmaceutical and biotechnology industries. Their mission is to help overcome challenges with innovative tools and solutions from discovery to the clinic. They supply life science tools designed to be used in discovery research and scalable to the clinical phase and beyond. By consistently adapting to new regulatory challenges and guidelines, ACROBiosystems delivers solutions, whether it comes through recombinant proteins, antibodies, assay kits, GMP-grade reagents, or custom services. ACROBiosystems empower scientists and engineers dedicated towards innovation to simplify and accelerate the development of new, better, and more affordable medicine.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.